The investigation related to quality-related events that occur from cGxPs approved specifications, and/or procedures including but not limited to Protocols, Master Batch Records (MBR), Batch Packaging Records (BPR), breakdowns, facilities, storage, distribution, manufacturing, testing, packaging, warehouse and distribution of drug products.
Guideline on Investigation Tools
1.0 PURPOSE:
-
- This Standard Operating Procedure (SOP) defines the requirements for reporting, documenting, investigating, evaluating, managing, resolving, and approving closure of investigation from cGxPs, approved specifications, and/or procedures.
2.0 SCOPE:
-
- This SOP/Guideline applies to Investigations related to quality-related events that occur from cGxPs, approved specifications, and/or procedures including but not limited to
-
-
- Protocols,
-
-
-
- Master Batch Records (MBR),
-
-
-
- Batch Packaging Records (BPR),
-
-
-
- Breakdowns,
-
-
-
- Facilities,
-
-
-
- Storage,
-
-
-
- Distribution,
-
-
-
- Manufacturing,
-
-
-
- Testing,
-
-
-
- Packaging,
-
-
-
- Warehouse and distribution of drug products.
-
3.0 REFERENCES – GUIDELINE FOR INVESTIGATION:
-
- SOP for Handling of Corrective and Preventive Actions (CAPA)…….<SOP>
-
- US FDA 21 CFR Part 211, 501, 600 and 820
-
- SOP for Quality Risk Management……….<SOP>
-
- European Commission: EudraLex – Volume 4 – Good Manufacturing Practice (GMP) Guidelines
-
- SOP for Handling of Market Complaints……..<SOP>
-
- ICH Q7 – Good Manufacturing Practice Guidance for Active Pharmaceutical Ingredients
-
- SOP for Deviations/Incidents
-
- CDER Guidance for Industry: Quality Systems Approach to Pharmaceutical
-
- cGMP Regulations, September 2006
-
- EMEA Guidance: Manufacture of finished dosage forms, April 1996
-
- SOP for Documents Management System
4.0 RESPONSIBILITY – INVESTIGATION TOOLS:
-
-
Quality Assurance Head/Designee shall be responsible for-
-
-
- The oversight and assessment of the investigation.
-
- Reviewing and approving the investigation plan, report, and timely resolution of all investigations.
-
- Assuring timely implementation of corrective actions and ensuring corrective actions are effective.
-
- Providing assistance in performing investigation.
-
- Reviewing and approving the investigation reports.
-
- Deriving appropriate CAPA and ensure adequate implementation of CAPA.
-
- Disposition of impacted products/batches and/or releases other controls, based upon investigation conclusions and associated corrections.
-
- Taking necessary action to notify customers or regulatory agencies about the outcomes of the investigations, wherever applicable.
-
- The overall resolution of all investigations originating from “Critical”/ “Major” events.
-
- Assuring timely implementation of corrective actions and ensuring corrective actions are effective.
-
- Reviewing and approving investigations impacting multiple sites and or requiring market action.
-
-
Manufacturing head/designee shall be responsible for-
-
-
- Ensuring all manufacturing deviations/incidents are reported to QA on the day of discovery, but no later than the end of the next working day.
-
- Ensuring that resources are available to support the investigation closure and assist QA.
-
-
Quality control head /designee shall be responsible for-
-
-
- Ensuring all deviations/incidents, non-conformances, failures, and/or events in the laboratory are reported to the QA on the day of discovery, but no later than the end of the next working day.
-
- Ensuring resources are available to support the investigation closure and assisting the QA as required.
5.0 ABBREVIATIONS – INVESTIGATION TOOLS:
-
- BMR: Batch manufacturing record.
-
- CAPA: Corrective and Preventive Action
-
- CCR: Change Control Record
-
- CFT: Cross-Functional Team
-
- EHS: Environmental health safety
-
- SME: Subject Matter Expert
-
-
DEFINITION:
-
-
-
Acceptance Criteria:
-
-
- The product specifications and acceptance/rejection criteria, such as acceptable quality level and unacceptable quality level, with an associated sampling plan, that are necessary for deciding to accept or reject a lot or batch (or any other convenient subgroups of manufactured units).
-
- The criteria, a system or process must attain to satisfy a test or other requirements.
-
- Assessment: The act or process, of evaluating (e.g. extent, magnitude, position, impact or compliance level) of a process, system, project, action, or activity.
-
-
Corrective and Preventive Action:
-
-
- A concept with current Good Manufacturing Practice (cGMP) that focuses on the systematic investigation of root causes of unexpected incidences to prevent their recurrence (corrective action) or to prevent their occurrence (preventive action).
-
- Corrective Action:
-
- Action is taken to eliminate the causes of an existing nonconformity, defect, or other undesirable situation, in order to prevent a recurrence.
-
- Preventative Action:
-
- Action is taken to eliminate the cause of a potential nonconformity, defect, or other undesirable situation, to prevent occurrence.
-
-
Cross-Functional Team:
-
-
- Diverse team members (SME) typically comprised of heads from Quality, Manufacturing, Qualified Person, Regulatory, Laboratory, or their qualified designees used to review and provide a disposition for the proposed deviation.
-
-
cGxP:
-
-
- cGxP is a general term that stands for current Good “x” Practice (x = Clinical, Engineering, Laboratory, Manufacturing, Documentation, Pharmaceutical, etc.).
-
- The titles of these Good “x” Practice guidelines usually begin with “Good” and end in “Practice”.
-
- cGxP represents the abbreviations of these titles where “x” a common symbol for a variable, represents the specific descriptor.
-
- Correction: Immediate action taken to resolve finding/issue.
-
-
Critical Classification:
-
-
- Any departure from established standards that has caused or has a high probability of causing adverse impact on product safety, quality, identity, potency, or purity.
-
-
Major Classification:
-
-
- Any departure from established standards which may have an impact on safety, quality, identity, potency or purity physical characteristics and efficacy of the product or process ( e.g. use of the different type of equipment).
-
-
Minor Classification:
-
-
- Any departure from established standards which may not have an impact on safety, quality, identity, potency, or purity physical characteristics and efficacy of the product or process ( e.g. use of the different size of a fiber drum).
-
- Deviation :
-
- Any departure (planned or unplanned) from approved procedures or records, including, but not limited to
-
-
- Standard Operating Procedure,
-
-
-
- Master Production Record,
-
-
-
- Batch Production Record,
-
-
-
- Standard Testing Procedure or the failure of a batch or any of its components to meet any of its specifications shall be documented and explained.
-
-
- Potential product quality impacting events shall be investigated and the investigation and its conclusions shall be documented.
-
-
Elimination:
-
-
- Errors Eliminate the possibility of error.
-
- This can be accomplished by eliminating the task.
-
- For example, eliminate mixing errors by purchasing pre-mixed materials.
-
- Eliminate recording errors by directly linking the measurement device to a printer.
-
- Event: Any unforeseen happening or unexpected occurrence.
-
-
Incident / Unplanned Deviation– Owner:
-
-
- An individual responsible for initiating the event/incident investigation determines the root cause and implements corrections.
-
- Incident/Unplanned Deviation:
-
- An unplanned or uncontrolled/unexpected GMP incident or deviation or an event in the form of departure from the designed systems or procedures at any stage of material receipt, manufacturing, packaging, testing, holding, and storage of drug substance and it is Intermediate/Components due to system failure or equipment breakdown or human interventions and observed at a later time during execution, audit, etc.
-
- Initiator – Incident/Unplanned Deviation: An individual who initiates an event/incident record.
-
- Initiator – Temporary Change / Planned Deviation:
-
- Functional Supervisor or the higher-level person who initiates a Temporary Change or a Planned Deviation record.
-
- Initiator- Investigation: An individual who initiates an investigation record.
-
- Investigation: A documented logical and/or scientific review of data related to all quality events that lead to the identification of the root cause and corrective and preventive action.
-
-
Market Action:
-
-
- An action required to resolve a situation for a marketed product.
-
- General reference embracing a notification, potential recall, market withdrawal, or field correction.
-
- Nonconforming Material: Material that does not meet specified acceptance criteria.
-
-
Product Quality Complaint :
-
-
- Any written, electronic or oral communication reported by Customers, Physicians, Pharmacists, Hospitals, Regulatory Agencies, Health Authorities, Government Laboratories, MA Holders, Retailers, Distributors, etc., that alleges deficiencies related to the safety, identity, strength, quality, purity reliability, and/or efficacy of a product after it is distributed beyond the control area quality systems.
-
- Subject Matter Expert: An individual, who is educated, trained, and/or highly experienced in a particular field or subject
-
-
Task:
-
-
- Any activity identified as a part of executing a ‘Temporary Change’ or ‘Planned Deviation’.
-
- Tasks can be of two types, i.e., pre-requi (to be completed before the execution of the temporary change or planned deviation) and non-pre-requi (can be completed concurrently with the execution of the temporary change or planned deviation).
7.0 PROCEDURE – INVESTIGATION TOOLS:
-
- An investigation may be required to determine root cause(s) for any of the following situations, but not limited to:
-
-
- Deviations/ Incidents/ Non-conformances/Failures/ Events.
-
-
-
- Product Quality Complaints
-
-
-
- Product Recalls, recovery, withdrawal, or return of goods.
-
-
-
- Internal/external audit or inspection findings.
-
-
-
- Periodic Product Quality Reviews.
-
-
-
- Ineffective Corrective and Preventive Actions (CAPA).
-
-
-
- Any other product quality or patient safety-related issues or review findings.
-
-
Initiation of investigations:
-
- An investigation record may be initiated by a Responsible Person from any department (referred to as “Initiator” hereafter) intending to investigate an identified quality issue.
-
- QA shall assign the investigation report number and make the necessary entry in the investigation number allocation log as per Annexure3.
-
- QA shall assign the investigation number as given below,
-
- The Investigation number shall be alphanumeric i.e. IR/XX/YY/ZZZ.
-
-
- Where,
-
-
-
- IR: Investigation Report
-
-
-
- XX: Department Code (Refer in Table -1)
-
-
-
- YY: Stand for the year. The last 2 digits of the year shall be 20 for 2020,
-
-
-
- ZZZ: Stand for serial no. of investigation which runs continuously irrespective of the change in department code.
-
-
- Every year 3 digit serial number shall start from 001.
-
- e.g. First Investigation report of 2020 in the Production department shall be numbered as IR/PR/20/001,
-
- Second Investigation report of 2020 in the QC department shall be numbered as IR/QC/20/002.
Table- 1
| Department | Code |
| Quality Assurance | QA |
| Quality Control | QC |
| Warehouse | WH |
| Production | PR |
| Maintenance | MN |
| Personnel & Administration | PA |
| Information Technology | IT |
| Environment, health, and safety | EHS |
| Formulation Development Department | FDD |
| Packaging Development Department | PDD |
-
- The Initiator shall initiate an investigation record and log the investigation number.
-
- The investigation record shall be submitted to the Responsible Person for further execution.
-
Investigation plan:
-
- The Responsible Person shall acknowledge and complete the hypotheses, investigation plan and approach, and obtain required resources (Materials, Equipment, Facilities, and Personnel) and required data (existing) and intended analytical techniques.
-
- If applicable, The Responsible Person shall attach supporting documents,
-
- The Responsible Person shall identify the cross-functional investigation team comprised of individuals from affected or other sites, resources external to the company, or Subject Matter Expert(s) in the area(s) relevant to the investigation
-
- The Responsible Person shall submit the investigation plan for approval to QA.
-
- QA may ask for additional information and/or request changes to the investigation plan.
-
- On a satisfactory review of the investigation plan, QA and Quality head shall approve the investigation plan.
-
- The cross-functional investigation team shall propose a hypothesis to identify the root cause(s) and consider the following elements during hypothesis development-
-
-
- Environmental monitoring excursions,
-
-
-
- Raw material non- conformance appropriateness,
-
-
-
- Process deviations – effectiveness,
-
-
-
- Equipment-instrument failure,
-
-
-
- Material or product mishandling,
-
-
-
- Storage condition excursion,
-
-
-
- Calibration excursions and non-conformances,
-
-
-
- Test method failure,
-
-
-
- cGxP non-compliances,
-
-
-
- Significant changes in historical frequency/trending of similar issues and prior actions are taken.
-
-
- The investigation team / CFT shall use appropriate investigative techniques to determine the root cause(s) as a part of the investigative approach.
-
- A list of potential techniques along with their applicability is provided in Table 1.
-
- This list is not meant to be exhaustive. See Annexure-1 for additional details for these tools.
Table 1: Investigation Technique
|
Objective of Analysis |
Possible Analysis Methods/Tools | Primary Use/Applicability |
Issue Analysis |
Affinity Diagram | A means to creatively generate a large number of ideas/issues and then organize and summarize them into natural groupings in order to understand the essence of a problem and breakthrough solutions. |
| Brainstorming | To creatively and efficiently generate a high volume of ideas on any topic. | |
| Prioritization Matrices |
To narrow down options through a systematic approach of comparing choices by selecting, weighting, and applying criteria. | |
Trend Analysis |
Check Sheet | To systematically record and compile data from historical sources, or observations as they occur so that patterns and trends can be clearly detected and shown. |
| Control Charts | To monitor, control, and improve process performance over time by studying variation and its source. | |
| Process Capability | To determine if a process, given its natural variation is capable of meeting established customer requirements or specifications. | |
| Run Chart | To study observed performance data for trends or patterns over a specified period of time. | |
Pattern Analysis |
Check Sheet | To systematically record and compile data from historical sources, or observations as they occur so that patterns and trends can be clearly detected and shown. |
| Flow Charts | To identify the actual flow or sequence of events in a process and to show the complexity, problem areas, redundancy, unnecessary loops, and places where simplification /standardization is possible | |
| Histograms | To summarize process data that has been
collected over a period of time and to graphically present its frequency distribution in bar form. |
|
| Matrix Diagram | To systematically identify, analyze, and rate the presence and strength of relationships between two or more sets of data. | |
| Normal Test Plots | To investigate whether process data exhibit the standard normal “bell curve” or Gaussian distribution. (Also, known as Normal Probability Plots, Normal Quartile Plots.) | |
| Pareto Charts | To focus efforts on the problems that offer the greatest potential for improvement by showing their relative frequency or size in a descending bar graph. | |
| Radar Chart | To visually show in one graphic the size of gaps among a number of both current organization performance areas and ideal performance areas. | |
Cause/Effect orLinkage Analysis |
Activity Network Diagram | To find both the most efficient path and a realistic schedule for the completion of any project. |
| Cause & Effect/Fishbone Diagram | To identify, explore, and graphically display, in increasing detail, all of the possible causes related to a problem and to discover its root cause(s). | |
Cause/Effect orLinkage Analysis |
Fault Tree Analysis or Failure Mode Effect Analysis | To analyze the effects of operating conditions on design reliability and safety. |
| Flow Charts | To identify the actual flow or sequence of events in a process and to show the complexity, problem areas, redundancy, unnecessary loops, and places where simplification/standardization is possible. | |
| Force Field Analysis | To identify the forces and factors in place that support or work against the solution of a problem, so that the positives can be reinforced and/or the negatives eliminated or reduced. | |
| Relations Diagram (or Interrelationship Digraph) | To systematically identify, analyze, and classify the cause and effect relationships that exist among all critical issues, so that key drivers or outcomes can become the center of an effective solution | |
| Scatter Diagram | To study and identify the possible relationships between the changes observed in two different sets of variables. | |
| Tree Diagram | To graphically representation and implement by taking a broad goal and breaking it into increasing levels of detailed actions that must be done to achieve the stated goals. | |
| 5-Why’s | To quickly separate symptoms from causes and identify the root cause of a problem. |
Note: Refer to Annexure 1 on “Tools for Root Cause Analysis” for more details.
-
-
The following program management-related activities shall be used during the investigation:
-
-
- Drive the investigation to the timeline in the plan; update the plan, as required, and maintain communication with functional management and other stakeholders on progress and issues.
-
- Identify and obtain resources from respective functional heads, as needed, to complete thorough and effective investigation.
-
-
- Escalate issues and roadblocks to management, as needed, when:
-
-
-
- Technical obstacles result in revisions to approach or timeline.
-
-
-
- Resources are not available to execute the plan.
-
-
-
- Functional alignment is not achieved on priorities.
-
-
Investigation Execution:
-
- The investigation team/CFT shall collect, organize, and document the data needed to perform a root-cause analysis.
-
- Data may come from a variety of sources, including, but not limited to the following, as given in Table 2:
Table 2: Possible Data Sources
| Potential Source of Issue | Possible Data Sources |
| General sources | Historical trends, including previously associated investigations, if applicable Interview of associated personnel Annual Product Quality Reviews Recalls/return goods |
| Environmental monitoring excursions | Microbiological test data Temperature and Humidity Monitorial data |
| Raw material nonconformance/appropriateness | Raw material receiving logs and records Raw material test procedure Raw material test reports |
| Process deviations/ effectiveness | Validation records Batch process records of affected batches and previously manufactured batches using the same equipment/room Change control history and log Stability data Process Package, Development Report On-line, at-line process monitoring/recording Systems Online cameras |
Equipment/instrument failure |
Preventive Maintenance and breakdown record Equipment/Instrument usage log Equipment /Instrument data printouts |
| Material/product mishandling | Packaging record(s) Shipping record(s) |
| Storage condition excursion | Storage facility/chamber monitoring record |
| Calibration excursions and non-conformance | Calibration records |
| Test method failure | Analytical test data Analytical method validation/verification report |
| GMP non-compliances | Personnel training records Deviation record |
-
- Conduct any additional experiments or tests, as required by the investigation plan to supplement data collected from existing sources.
-
- If the investigation is for any findings of contaminant, physical, biological, or chemical in nature, it shall be confirmed by analysis for its nature and the extent of impact shall be determined with a detailed investigation.
-
- The suspected causes or hypotheses shall be verified with adequate experiments, simulation processes, or backed up with strong, supporting scientific rationale to assign as the root cause.
-
- Identify other investigations that were similar in nature and review the investigation approach and results of those investigations as a rationale to assign root cause.
-
- Perform an impact assessment and extend the investigation to other batches and other products of all the markets, which could have been possibly affected.
-
- The investigation may include additional testing of the involved batches of components.
-
Analysis and conclusion:
-
- The investigation team/CFT shall assess the data collected during the investigation and document the root cause(s).
-
- An appropriate investigative technique, as listed in Table 1, may be used for analysis, if needed.
-
- The evaluation shall be performed as indicated below.
Note: that the steps listed below are indicative of the types of questions to ask; these do not constitute a comprehensive list. Attach the supporting data, if any.
-
-
Evaluation of GMP Compliance:
-
-
- Confirm processes were followed (i.e., no operator error was found).
-
- Confirm process expectations were understood (i.e., no subjectivity exists as part of the process; operator understands what to do at each stage of the process).
-
- Review completed documentation, Batch records, logbooks, etc., to confirm appropriate data was captured/analyzed by the operator.
-
- Evaluation of Equipment and Facilities.
-
- Determine how equipment usage may have contributed to the issue.
-
- Review qualified parameters to determine if running at process extremes may have contributed to the issue.
-
- Confirm set-up, shut-down, and cleaning processes were followed.
-
- Review maintenance records to determine if recent maintenance may have contributed to the issue.
-
- Check the calibration requirements and record to confirm the machine are currently calibrated.
-
- Review spare parts / interchangeable parts to determine if parts failure may have contributed to the issue.
-
- Consult with machine vendors/equipment manufacturers/technical experts, as appropriate.
-
-
Evaluation of Process:
-
-
- Determine if the process was properly validated.
-
- Determine whether the process is fully and clearly documented to facilitate unambiguous understanding and interpretation by the intended user.
-
- Assess the possibility of any weakness (lack of robustness) or errors in the process.
-
- Ensure that process documentation forms, including batch process records are correct and complete.
-
-
Evaluation of Analytical/Microbiological Method:
-
-
- Determine if the method was properly validated/verified.
-
- Verify that the method is fully and clearly documented to facilitate unambiguous understanding and interpretation by the intended user.
-
- Assess the possibility of any weaknesses (lack of robustness) or errors in the method.
-
- Assess the possibility of ambiguity or error in data analysis associated with the method.
-
-
Evaluation of Storage/Handling:
-
-
- Determine whether documented storage and handling requirements are appropriate and have been met.
-
- Assess whether other batches were subjected to the same storage and handling conditions and whether adverse effects were identified on other batches also.
-
-
Evaluation of Material:
-
-
- Review incoming material test data; confirm raw material specifications have been met.
-
- Identify if material lot/batch was used on other products, identify if adverse effects were identified on other product batches.
-
- Confirm material identification.
-
- Consult with vendor/material manufacturer, as appropriate.
-
-
Others:
-
-
- Determine if abnormal manufacturing variations or deviations or incidents occurred on the line, such as (but not limited to) power spikes or outages; extreme environmental conditions, accidents, spills, or safety
-
- The investigation team/CFT shall conclude the investigation based on the data analysis and categorize the root cause(s) into one of the categories in Table 3:
Table 3: Root-Cause Categories
| No. | Root Cause category |
| 1. | Lab Instrument Inadequacy |
| 2. | Lab Instrument Malfunctioning |
| 3. | Lab Instrument Maintenance |
| 4. | Lab Instrument Software |
| 5. | Lab Testing Method |
| 6. | Equipment Malfunctioning |
| 7. | Facility/Utility Malfunctioning |
| 8. | Equipment Inadequacy |
| 9. | Facility/Utility Inadequacy |
| 10. | Facility/Utility Software |
| 11. | Equipment Software |
| 12. | Equipment Maintenance |
| 13. | Facility Maintenance |
| 14. | Utility Maintenance |
| 15. | Inadequate Document Control |
| 16. | Product Development |
| 17. | Packaging Development |
| 18. | Labeling |
| 19. | Manufacturing Process Validation |
| 20. | Raw Material |
| 21. | Procedural Inadequacy |
| 22. | Packaging Material |
| 23. | Procedural Non-Compliance |
| 24. | Storage |
| 25. | Specification/Standard Test Procedure (STP) Inadequacy |
| 26. | Transportation & Handling |
| 27. | Planning |
| 28. | Ineffective Training Program |
| 29. | Sampling |
| 30. | Sample Handling |
| 31. | Others – specify in the “Root Cause Summary” |
-
- Upon completion of the investigation, if the root cause(s) cannot be identified, then the most probable cause(s) shall be provided by the investigation team/CFT.
-
- Details of the investigation and the analysis performed shall be documented.
-
- All raw data and supporting investigation files shall be archived, as per applicable procedure in accordance with the current version of the respective SOP.
-
-
Investigation reports shall have the following content, but not limited to:
-
-
- Cover Page:
-
-
- The cover page shall include
-
-
-
- Title,
-
-
-
- Location,
-
-
-
- Unique Investigation Number,
-
-
-
- Page number, and Company Logo.
-
-
- This information shall be reflected on all the pages of the investigation report.
-
- Table of Contents Page:
-
- The table of contents page shall include the content of the investigation with the respective page numbers.
-
- Approval Page:
-
- The approval page shall include “prepared by”, “reviewed by” and “approved by” signatory columns.
-
- The report shall be prepared by the Investigation Owner in coordination with a cross-functional team of investigation.
-
- The report shall be reviewed by the cross-functional team or QA and finally approved by Head QA/designee and Quality Head.
-
- Objective:
-
- The objective shall include the reason for the investigation, assessment of impact/risk, and explore the CAPA to mitigate the risk.
-
- Background:
-
- The background shall constitute short summary concerning initiating the investigation based on the master document.
-
- Scope:
-
- The scope shall include the reference to the product name and reference number of the Deviation/Incident/Market compliant/Product recall of the respective product.
-
- Responsibility:
-
- The Responsibility shall cover the name of the individual(s) involved in the cross-functional investigation team/CFT and their responsibility during the investigation.
-
- Investigation Results and Discussion:
-
- Summary observations, data ranges, defects, deviations, or incidents shall be included in this section, and all investigation results shall be summarized.
-
- Additional Experimental Data:
-
- The data of additional experiments conducted shall be included and a conclusion documented.
-
-
Root Cause/Most Probable Root Cause:
-
-
- List the root cause by providing the reference documents’ numbers which support sound scientific List the most probable root-cause when an investigation is not conclusive.
-
- Describe the specific basis for ruling out possible root causes along with reference numbers to supporting documents.
-
-
Impact Assessment:
-
-
- List all the batches/products/ systems/ procedures impacted by the failure(s).
-
- Impact Assessment shall be assessed with respect to patient safety, product quality, other batches/product, regulatory commitments, and business.
-
- Risk Assessment: List the risk identified and proposal for reduction or elimination of the risk.
-
- CAPA: Provide CAPA details.
-
- Conclusion:
-
- List the conclusion.
-
- The conclusion shall be supported by documented evidence based on sound scientific principles, logic, experimental data, and all other relevant factors.
-
- List of Annexure:
-
- Provide the annexure number for each of the supporting documents referred to as a part of the investigation.
-
Investigation closure:
-
- Complete the investigation summary:
-
- Recommended CAPAs; potential controls to assess, mitigate, and reduce the risk of recurrence.
-
- Include potential risks identified as an outcome of the investigation.
-
- Provide the process for communicating risk to impacted areas.
-
- Include the proposed plan for monitoring risk moving forward.
-
- Ensure any open commitments that cannot be completed before the investigation is closed are being regularly tracked to ensure they are completed by their respective deadline(s).
-
- Investigations shall be completed within thirty (30) calendar days from the time that the quality event is first discovered.
-
- If an investigation cannot be completed within the required time frames, the Responsible person shall request an extension from QA with the proposed new timeline and justification for the extension request.
-
- The request for extension shall also include an impact assessment to evaluate the effect on activities still to be completed through the completion of the investigation.
-
- This assessment shall determine if additional controls are needed to avoid issues during the interim.
-
- This justification for the extension of timelines shall be reviewed and approved by QA prior to implementation.
-
- If an investigation cannot be completed within the required time frames,
-
-
The Responsible Person must submit an interim report at least every 30 days for the same.
-
-
- The investigation record shall be submitted to the QA and as applicable, to determine adequacy, completeness, and approval of remedial actions required.
-
- QA as applicable may ask for additional information and/or ask to perform additional work as a part of the investigation.
-
- On satisfactory completion of the investigation record, QA shall complete a risk classification (Critical, Major, and Minor) along with documentation of the issue that led to the investigation and finally close the record.
-
- Each investigation report shall be approved by the Head QA/designee.
-
- Types of Investigations that may require escalation to the Management in accordance with the written procedures as per the current version of the respective SOP.
-
-
- Investigations “OPEN” for more than 30 calendar days.
-
-
-
- In which Investigations there is no assignable root cause but only a most probable root cause.
-
-
-
- Investigations concluding into product stoppage/manufacturing hold,
-
-
-
- Investigation concluding into product/batch recall
-
-
- QA shall disposition impacted products/batches and/or releases other controls, based upon investigation conclusions and associated corrections.
-
- After the closure of the investigation, if further investigation is required (e.g. due to request by QA, discovery of new information/data, internal/external audit observations, or management review), closed investigations shall be re-opened by the Quality Head/designee or Responsible Person to perform and document the further investigation.
-
- The implementation and effectiveness of CAPA shall be monitored as per written procedures.
-
Requirements:
-
- Each employee is responsible for the identification of deviation/incident/non-conformance/failures/events at the time it occurs and reporting to the supervisor immediately.
-
- Establish and maintain a system to assure the investigation is conducted by a Responsible Person (RP) in conjunction with QA Head or designee and other involved functions, as required.
-
-
The investigation shall include the following information:
-
-
-
- The description shall include an objective, factual description based on the science of what happened and when.
-
-
-
- Immediate action is taken, the decisions made about the process and product affected shall be documented.
-
-
-
- The scope shall include an assessment concerning when the process, equipment, facility, utility, or other source was the last working properly.
-
-
-
All known concerns, not limited to the following, shall be included in the report:
-
-
-
- Patient or worker safety.
-
-
-
- Out-of-Specification and Out-of-Trend observations.
-
-
-
- Compliance with cGxPs, compliance with registration, deviation/incident from process validation.
-
-
-
- Impact on product safety or efficacy.
-
-
-
- Product stability impact.
-
-
-
- Effectiveness of previous root cause corrective actions.
-
-
-
- Notification requirements.
-
-
-
The gravity of the concern(s) shall be determined by assessing how significant the impact is.
-
-
-
- Any additional concern(s) identified during the course of the investigation shall be documented.
-
-
-
- The resolution of each concern shall be described, and any unresolved concerns shall be clearly identified.
-
-
-
- The impact on the status of the lot shall be described.
-
-
-
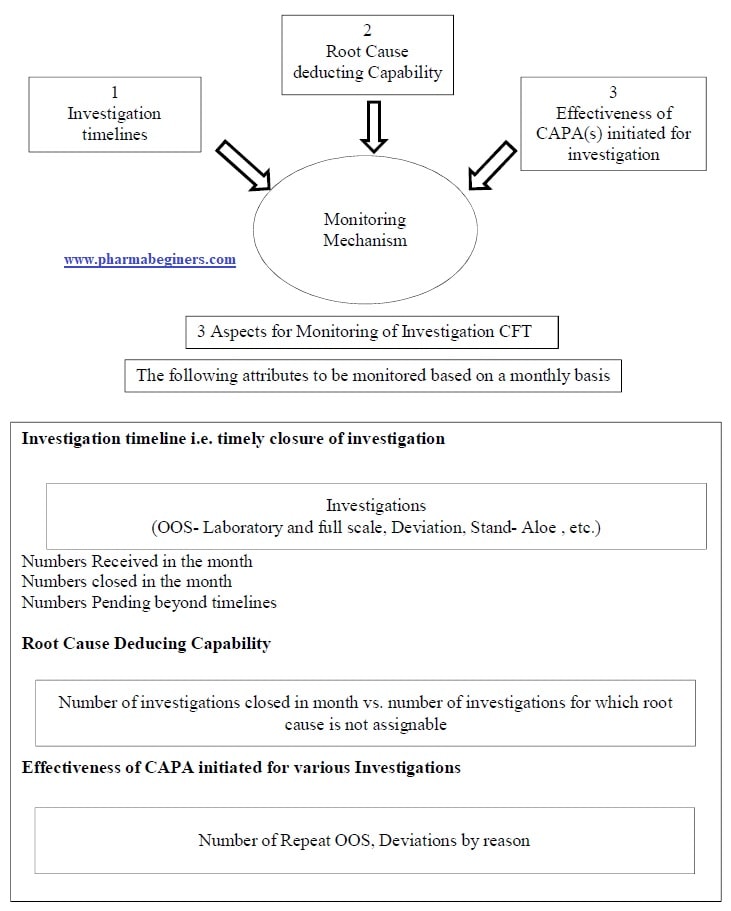
- The monitoring mechanism for investigations by the CFT shall be developed, refer to Annexure -2 for a schematic diagram.
-
-
Root cause investigation:
-
- The most probable cause of the deviation/ incident/ non-conformance/failure/ event and what was done to confirm the true cause shall be described.
-
- The investigation shall include the causes that were specifically eliminated as the root cause and supporting rationale for that judgment shall be documented.
-
- The basis of the cause shall be described.
-
- Corrective actions taken to address the issue shall be described.
-
- Corrective and Preventive Actions (CAPA) taken and planned shall be described and tracked through completion.
-
- Lot Corrective Action: A description of the corrective action for the lot (e.g. reprocessing, rework, downstream processing, inspection, culling, destruction, restriction) and similar incident history shall be provided, if applicable.
8.0 ANNEXURES:
Annexure 1: Investigation Report Template
INVESTIGATION REPORT OF (Title)
| Date of deviation/Incidents: | |
| Reference Deviation/Incident No.: | |
| Category of Deviation/Incident: | |
| Discrepancy details | |
| Product/Material/ System Name: | |
| Batch No./ Document No./Equipment ID No.: | |
| Targeted completion date | |
| Revised Target date: | |
| Date of Closing | |
| Closing Status |
- TABLE OF CONTENT
| Sr. No. | Content | Page No. |
| 1.0 | Table of Content | |
| 2.0 | Approval Page | |
| 3.0 | Objective | |
| 4.0 | Background | |
| 5.0 | Scope | |
| 6.0 | Responsibility | |
| 7.0 | Investigation Results and Discussion | |
| 8.0 | Additional Experimental Data | |
| 9.0 | Root Cause/Most Probable Root Cause | |
| 10.0 | Impact Assessment | |
| 11.0 | Risk Assessment | |
| 12.0 | CAPA | |
| 13.0 | Conclusion | |
| 14.0 | List of Annexures | |
| 15.0 | Closure of Investigation |
Annexure 2: Monitoring Mechanism for Investigation CFT
Annexure 3: Investigation Initiation
| Discrepancy Discovered on: | ||
| Reported By: | ||
| Discrepancy Details
|
||
| Prepared by
Sign & date |
Checked by Dept. Head
Sign & date |
|
Annexure 4: Investigation Log
| Sr.
No.
|
Investigation No. | Product / material / equipment / instrument / system name | Doc. No. / B. No. / Equipment No./ Instrument No. | Investigation description | Ref. Event Report No. | Target Date of Completion |
| Extension Date of investigation | Responsible person | Status
(open /closed). |
CAPA No. | Remark | Sign |
Annexure 5: Tool for Root Cause Analysis
Click to download Tool for Root Cause Analysis
Annexure 6: Investigation Extension Request
| Investigation No. | Date of Initiation | |||||
| Reference No. | ||||||
| Details for investigation | ||||||
| Initial Target Date | Proposed Target Date | |||||
| Justification of Extension/Delay: (Attach the objective evidence, if any) | ||||||
| Prepared By | Comments | Sign & Date | ||||
| HOD | ||||||
| QA Head | ||||||
| Plant Head | ||||||
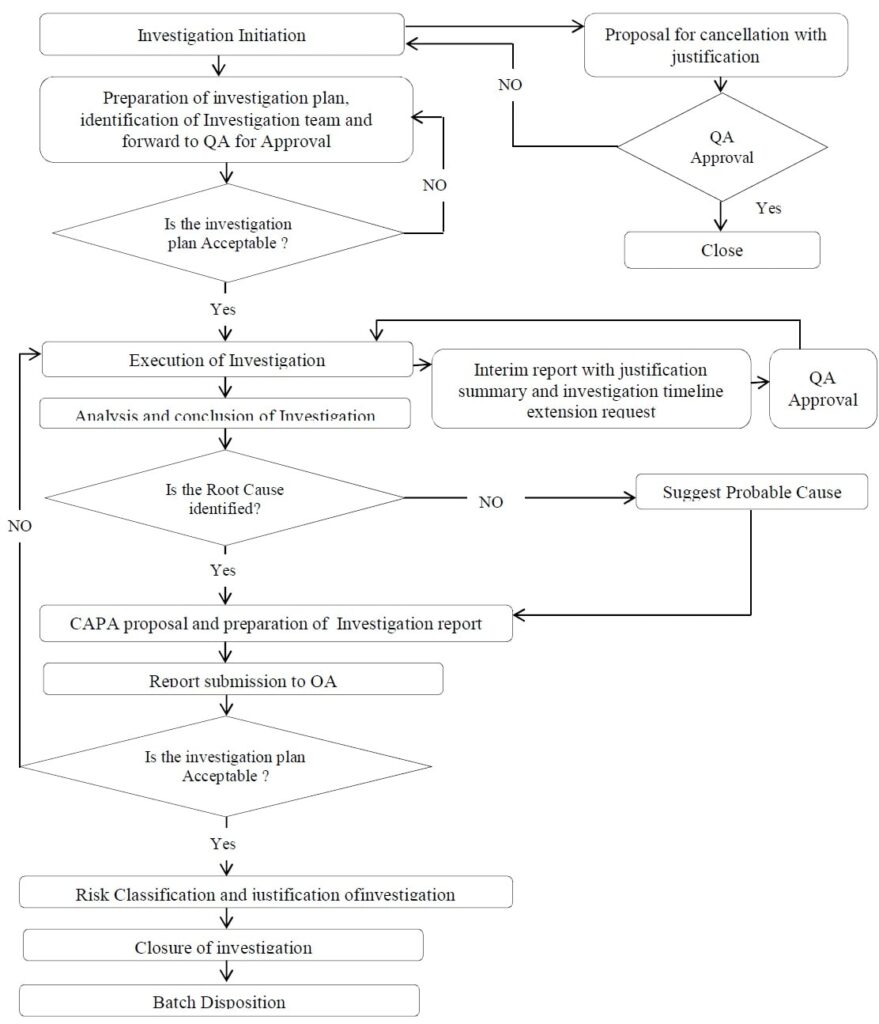
Annexure 7: Work Flow for Investigation
**********************************************END**********************************************



Pingback: Environmental Monitoring Guide - Non Sterile Facility - Pharma Beginners