In the SOP on Internal Audit and Self Inspection, describe the detailed procedure to carryout the the Self inspection and Internal Audit in various department of pharmaceuticals.
Audit is a systemic and independent examination to determine the quality activities and related results comply with planned arrangements and whether these arrangements are implemented effectively and are suitable to achieve objectives.
SOP for Internal Audit and Self Inspection
Purpose:
- To define procedure for carrying out internal audits and Self inspection in various department in pharmaceutical plant
- For systemic examination of quality systems and processes.
- To verify compliance with applicable regulatory cGMP requirements,
- To improve system and to determine the effectiveness in meeting specified objectives.
Scope :
- This SOP is applicable to Internal audit and Self Inspection carried out in the below listed departments (but not limited to) :
- Warehouse
- Manufacturing (Granulation, Compression ,Coating & Capsulation, Filling etc.)
- Packing
- Personnel & Administration
- Quality Control
- Maintenance
- Quality Assurance
- Environment Health and Safety (EHS)
- This SOP is also applicable in case of unscheduled internal audits carried out based on any deviations, event, market complaint, external audit observations and if any other case by the operating team of location at location.
References & Annexures:
-
References:
- Schedule M (Drug and Cosmetic Act 1945)
- WHO TRS 961
-
Annexures:
- Internal Audit
- Schedule (Annexure – 1)
- Intimation Form (Annexure – 2)
- Execution Form (Annexure – 3)
- Observation Report (Annexure – 4)
- Compliance cum Closing Report (Annexure – 5)
- Checklist (Annexure – 6)
- Warehouse Checklist (Annexure – 7)
- Manufacturing Checklist (Annexure – 8)
- Packing Checklist (Annexure – 9)
- Quality Control Checklist (Annexure – 10)
- Engineering/Utilities Checklist (Annexure – 11)
- Personnel & Administration Checklist (Annexure – 12)
- Quality Assurance Checklist (Annexure – 13)
- Certificate of Qualified Auditors for Internal Audit (Annexure – 14)
- List of Certified Auditors for Internal Audit (Annexure – 15)
- Internal Audit Log Book (Annexure – 16)
- Internal Audit
Responsibilities :
-
Auditee Department (Where Internal Audit and Self Inspection to perform) :
- Inform relevant employees about the objective and agenda of the audit.
- Appoint responsible person of staff to accompany audit team.
- Provide all resources needed for the audit team in order to ensure an effective and efficient audit process.
- Provide access to the facilities and evidential material as requested by the auditors.
- Cooperate with the auditors to permit the audit objectives to achieve.
- Determine and initiate core action based on the audit findings.
-
Auditee : Head Of Department:
- Ensure compliance in proper way.
- Guide staff to prepare / compile the compliance report.
- Ensure that audit compliance report is submitted within the stipulated time-lines as given in the SOP.
-
Auditor for Internal Audit or Self Inspection:
- Prepare a checklist for the audit.
- Plan and carry out audit.
- Impart training if required at the time of concluding meeting.
- Collect evidences and gather information through interviews.
- Prepare audit observation report based on audit findings and send to auditee.
-
Quality Assurance responsibilities during Internal Audit and Self Inspection:
- Organize for the audits and to prepare and approve yearly audit schedule covering all the departments.
- Follow up for the compliance of non- conformance as per the audit observation report.
- Review audit observations report and compliance report.
- Retain and safeguard the documents pertaining to the audit.
- Decide the auditor(s)/ audit team in consultation with quality/ factory head.
- Provide prior information to auditee and auditor.
- Issue photocopy of certificate to an auditor and retain original copy.
- Monitor the open observations.
Also read: SOP for Audit Trail Review and Privilege Policy
-
- Close audit compliance report.
- Generate and maintain the internal audit log.
- Initiate scheduled/unscheduled audit.
- Maintain the list of certified auditors.
-
Quality Head responsibilities during Internal Audit and Self Inspection:
- Authorize for the re-scheduling of planned audit dates.
- Approve internal audit schedule, audit observation report and certificate of auditors.
- Initiate out of schedule audit.
- Give approval for providing internal audit reports to FDA or any other agency.
- Supervise the regular internal audits or self inspection.
-
Operation Head or Plant Head :
- To give approval for providing internal audit reports to FDA or any other agency.
Distribution of SOP on Internal Audit and Self Inspection:
- Quality Assurance
- Quality Control
- Production
- Packing
- Warehouse
- Personnel and Administration
- Maintenance
Abbreviations & Definitions of Terms:
-
Abbreviations:
- CDER : Center for Drug Evaluation and Research
- cGMP : Current Good Manufacturing Practices
- EU GMP : European Union – Good Manufacturing Practices
- EHS : Environment, Health & Safety
- Code : Employee Code
- GMP : Good Manufacturing Practice
- Mfg : Manufacturing
- MN : Maintenance
- P & A : Personal & Administration
- Pkg : Packing
- QA : Quality Assurance
- QC : Quality Control.
- SISPQ : Safety, Identity, Strength, Purity and Quality
- US FDA : United State Food and Drug Administration
- WH : Warehouse
-
Definition of Terms:
-
What is the role of an internal audit?
- A systemic and independent examination to determine whether quality activities and related results comply with planned arrangements and,
- whether these arrangements are implemented effectively and are suitable to achieve objectives.
- Auditor (Audit team):
- The person or a group of persons performing audits.
- Auditee:
- The proposed department/ section/ area to audit.
- Audit Team:
- A chosen group of individuals from one or more departments with specific skill sets, job experiences, education or any combination there of who are properly prepared to conduct audits.
- The team members must have a good understanding of cGMP and laboratory procedures and display professional conduct at all times. .
- Observation:
- A statement of fact made during an audit and substantiated by objective evidence, observation shall be classified as mentioned below:
- Minor:
- No impact on SISPQ of product.
- Major:
- General established system deviations that have potential impact on SISPQ of the product.
- Critical:
- System deviations that have confirmed impact on SISPQ of the product and may cause product recalls and patient’s health risk.
- Nonconformity:
- The non-fulfillment of specified requirements.
-
Procedure (SOP on Internal Audit and Self Inspection):
- Audit Planning:
- Quality Assurance department shall prepare internal audit schedule annually covering all departments as per Annexure -1.
- Each department (Sub-section) shall audited once in a year.
- Adopt the vertical and horizontal approach during audit.
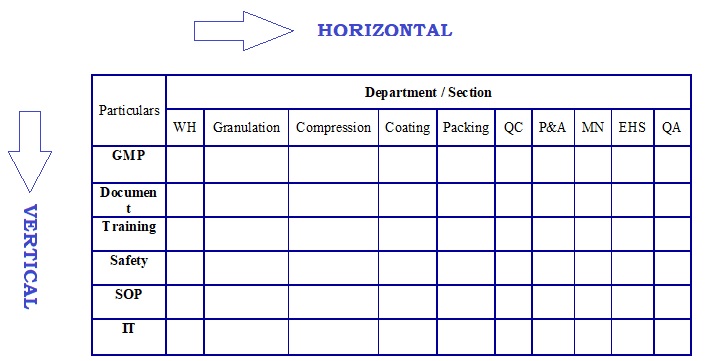
-
Horizontal approach shall specific/occasional/one time in all sections/unscheduled.
- For Example, Based on event, internal audit shall carry out. In this audit, horizontal approach shall adopt.
- For example, Audit related to Training shall carry out in all sections. This is horizontal, one time in all sections.
-
Vertical audit approach shall take for doing audit on overall activities of the particular section/department.
- For Example, Scheduled audit shall take as vertical approach of audit.
- During this type of audit, all particulars related to activity of auditee section/department, particulars related to GMP, safety and IT, particulars related to training etc. shall audit.
- Internal audit shall also carry out. Out of schedule on instruction of QA / Quality Head (if felt necessary). based on event, market complaint, external audit observations, OOS and repetitive failures at location.
- First week of every month (Self Inspection Schedule) , QA designee shall forward soft copy of internal audit intimation form through mail to the auditors and auditee as per schedule to confirm mutually convenient date for audit.
- Hard copy of internal audit intimation form which shall send through mail for approving response on date of audit as per Annexure-2 from auditors and auditee shall retain to QA with communication mail.
- Auditors and auditee shall give their response as per Annexure-2 (Soft copy) through mail to QA. Hard copy shall retain to QA with communication mail.
- On receipt of confirmation from both auditors and auditee, audit date shall finalize.
- If auditor and auditee are not able to finalize date for audit, QA Head / Quality Head shall intervene and date shall finalize.
-
Audit can advanced or postponed by 1 month in following situations :
- The Audit area is under shut down during the proposed period of audit.
- The proposed period is having a regulatory audit schedule.
- Proposed period is inconvenient to auditee due to other commitments.
- Advancing or postponing the audit dates in such case shall be done only after authorization of QA Head.
- New decided date shall intimate by auditee to QA as per Annexure-2.
- QA shall put their comments as per Annexure-2.
-
Selection of Auditors / Audit team :
- Management shall appoint a self-inspection team consisting of experts in their respective fields and familiar with GMP. The members of the team may appoint from inside the company.
- The team responsible for self-inspection shall consist of personnel who can evaluate the implementation of GMP objectively.
- Management shall constitute a team of independent, experienced, qualified persons from within the company, who can audit objectively the implementation of methodology and procedures evolved.
- For an audit in a particular department, certified auditors shall selected.
-
Auditors shall selected on the basis of following criteria.
- Person selected for audit shall not from the same department.
- The person shall have experience of working on dosage form similar to that of auditee department.
- Person (Auditor) shall have minimum experience of 1 year in the company.
- However, in specific cases, Quality Head / QA Head can over rule this requirement based on previous experience of the person.
- Ensure that person has trained on SOP of Internal Audit.
- Person is aware of internal audit system and has faced an audit (Internal Audit or Regulatory audit) as :
- An auditee in a Lead role had faced audits in support role.
- Has been an observer of such audit.
- Issue the photocopy of original certificate to an auditor who fulfills the above prerequisite as per Annexure-6 and,
- Original certificate shall retain with QA. QA shall maintain the list of certified auditors in excel sheet as per Annexure-8.
- Auditor(s) / audit team shall select as per the schedule prepared in the beginning of year.
- In case of emergencies or vacations; auditors can changed by QA Head.
- The name of alternate auditor shall mention in internal audit report in Name of auditors’ column.
- List of certified auditors shall prepare in excel sheet annually at the time of preparation of Internal Audit Schedule.
- Print out of list of certified auditors shall take and kept along with Internal Audit Schedule.
-
Intimation of Audit to the Auditors and Auditee :
- Audit shall announce by QA one week prior to the audit.
- An Audit Intimation Form (Annexure-2) shall circulate to auditors, auditee and Quality/Factory head which shall contain the following details:
- Audit Objective: Mention about the aim/purpose of the audit.
- Audit Agenda: Mention briefly about the areas, systems, SOPs & documentation part that are going to cover during the audit.
- Mention about name of the auditee department.
- Team (Auditors): Name, department/section of the auditor shall mention.
- Date of Audit: Mention date of audit.
-
Executing the Audit :
- At the time of execution of the audit, QA shall allocate number for self-inspection and make entry in Self-inspection Log book (Annexure-9 of SOP on Internal Audit and Self Inspection).
- At the time of execution of the audit, Internal Audit Execution Form (Annexure-3) shall fill by all auditors and auditee who shall participate in internal audit of that particular date.
- Hard copy of Annexure-3 (of SOP on Internal Audit and Self Inspection) shall retain to QA.
- Internal Audit (Self Inspection) numbering system:
SI-YY/DC-XXX,
Where,
SI : Self Inspection,
YY : Last two digits of the current year,
DC : Department Code,
XXX : Serial no. of audit performed.
- Auditors shall discuss the approach to be adopted for conducting an audit. A checklist (Annexure-6 of SOP on Internal Audit and Self Inspection) shall prepare for conducting an audit.
- Written instructions for self-inspection shall establish to provide a minimum and uniform standard of requirements.
-
Written instructions for self-inspection shall draw which shall include the following:
- Personnel
- Premises including personal facilities
- Maintenance of buildings and equipment
- Storage of starting materials and finished products
- Equipment
- Production and in-process controls
- Quality Control
- Documentation
- Sanitation and Hygiene
- Validation and re-validation programs
- Calibration of instruments or measurement systems
- Recall Procedures
- Complaints management
- Labels Control
- Results of previous self-inspections and any corrective steps taken.
- Auditors shall have an opening meeting with auditee and other supporting team members and explain the purpose of audit and procedure being followed for the audit.
- Auditors shall perform the audit. During audit, auditors shall try to cover all the area and systems being followed in the department for audit.
-
Auditors shall –
- Free from bias and influences, which could affect objectivity.
- Report critical nonconformity to the auditee or audit team on the spot.
- Report obstacles, if any, encountered in performing the audit.
- Remain within the audit scope.
- Exercise objectivity. As far as possible objective evidence from the shop floor shall be collected.
- For traceability of observations of nonconformity, mention the details of Product name, batch no., reference document no., page no., line no., date, name of the person.
- At the end of audit, in closing meeting, auditor(s) shall discuss with the auditee/department head about the audit findings (Eg: Nonconformity, the areas of concern, good practices that are followed) in presence of all concerned staff of the department & QA Head.
- If required, training shall provided by auditor to all the concerned persons & record of the same shall maintained.
-
Collection of evidence during Audit:
- Evidences shall collect through interviews, examination of documents, and observation of activities and conditions in the areas of concern.
- Clues suggesting nonconformity shall note as they seem significant, even though not covered by any documents and shall investigate.
- Information gathered through interviews may test by acquiring the same information from other independent sources, such as physical observation, measurements and records.
- In case of any difference of opinion between the internal auditors and auditee, matter shall be referred to QA Head and decision of QA Head shall be acceptable.
-
Audit Report Preparation:
- A report shall made at the completion of a self-inspection. The report shall include:
- Self-inspection (Internal Quality Audit) results.
- Evaluation and conclusions
- Recommended corrective actions
- A report shall made at the completion of a self-inspection. The report shall include:
-
After the completion of the audit,
- The auditor(s) / audit team shall review all of their observations to determine which are to be reported as nonconformity.
- The auditor(s) / audit team shall ensure that these are documented in a clear concise manner and are supported by the evidence.
- Nonconformities shall identify in terms of specific requirement of the standard or related documents against which the audit has been conducted.
- All auditors involved in the audit shall prepare an audit report of observations of non – conformity as per Annexure-4 (SOP on Internal Audit and Self Inspection) within 15 calendar days after the audit.
- The auditor shall also mention the satisfactory findings in the audit report.
Note : All the observations shall be documented. Recommendation shall be given where ever required.
- The audit report shall prepare in such a way so that it gives clear understanding of the content/observation where ever applicable.
- For example, it shall contain the details but not limited to the following i.e product name, batch no., reference document no., page no., date & name of the person.
- Auditor shall prepare Audit observation report based on audit findings.
- All observation shall be classified as Minor, Major or Critical. Critical observation shall inform within one working day to the auditee, plant head, and quality head during the course of audit which will enable auditee to comply critical observation as per the cGMP requirements.
- After approval of the Audit Report, the critical and major audit findings along with corrective & preventive actions, as applicable,
- Compliance to the critical observations shall be done as soon as possible or addressed within 5 working days and same shall report in the audit compliance report.
- Auditor shall mention as “Response is not required” for the observations those are in compliance with SOP / system being followed.
- Audit report to be send to Auditee for compliance of non-conformity.
- Auditor shall also brief QA Head about the audit findings.
- QA shall review the audit report and get the event raised, in case of any critical observation.
- QA shall send soft copy of internal audit observation report to auditee department through mail and approved hard copy shall be kept with QA.
-
Compliance Report Preparation and Closing Of Audit Report :
- Auditee shall produce a compliance report and its compliance status as per Annexure-6.
- Action plan with Target date of completion and name of the Responsible person shall mention for all open observation and
- Report shall forward to Auditors.
- All observation shall have two level closures. One for corrective action/disposition and another is for preventive action (long term) across as applicable.
- Auditors shall verify the compliance report and shall give concurrence for the action plan for compliance.
- If the action plan is not acceptable the report shall be sent back to auditee for rectification.
- Compliance report along with all supporting documents shall be submitted to QA within 30 calendar days from the date of receipt of audit observation report.
- Any deviation from this time-line shall be subject to prior approval from Quality Head or designee.
- QA shall verify the compliance closure.
- Once all the audit observations are closed, QA designee shall sign off the report for closure.
Note : QA shall monitor the open observations by keeping respective reports open.
(Refer Checklist in SOP on Internal Audit and Self Inspection)
- Audit report shall close once all the observations are complied.
- Auditor may decide for a follow up audit for confirmation of compliance.
- QA Head or Quality Head may also instruct auditors for a follow up audit for confirmation of compliance of certain critical observations.
- The Internal Audit Log shall generate and maintain by the QA.
- Auditee shall request with justification for an extension of target completion date in case of any variation.
- QA Head or Quality Head can extend the Target Date of Completion if found justified.
- Revised Target Date shall be mentioned in compliance report (action plan) manually after striking off the old target completion date.
- If there is any constraint in the closure of the audit report, the same shall be discussed with Corporate Quality and Management.
Note: If FDA or any other agency asks internal audit report then upon approval of Quality Head and Plant Head those shall be given.
For Attachment and Formats of above mentioned SOP, > Click here :
Also Visit : Checklist and Formats for Internal Audit and Self Inspection



Pingback: Cleaning Validation master plan (CVMP)-New Approach - Pharma Beginners
Pingback: Regulatory Audit : Preparation & Handling - Pharma Beginners
Pingback: Audit Checklist - Department wise in Pharmaceuticals - Pharma Beginners
Pingback: Site Master File (SMF) Preparation SOP & Guideline - Pharma Beginners
Pingback: Good Laboratory Practices (GLP) - SOP & Guideline - Pharma Beginners
Pingback: Internal Audit (Self Inspection) Checklist and Format :Pharma Beginners
Pingback: Internal Audit (Self Inspection) Checklist and Format :Pharma Beginners