Out of trend (OOT), results are those results that are within the specification but indicate that the particular analysis or process could be out of control.
In case of stability out of trend (OOT) results describes as “A results that are within the specification but do not follow the trend, with respect to previous station results during stability study”.
SOP for Handling Out of Trend (OOT) Results
1.0 Purpose:
-
- To provide the guidance/procedure for the handling of out of trend (OOT) results observed in quality control departments of drug substance & drug product manufacturing location.
2.0 Scope:
-
- This SOP shall be applicable for the following stage samples analyzed at pharmaceutical manufacturing plants.
-
- Analysis where additional testing criteria are given in pharmacopeia e.g. Dissolution & Content Uniformity Test.
-
- Out of Trend (OOT) results generated in Finished Products, Raw materials, Any other stage samples, and Stability Samples.
3.0 Reference, Attachments for Handling of Out of Trend (OOT) Results :
-
-
References :
-
-
- Pharmaceutical technology: Identification of out of trend stability results, PhRMA CMC statistics, stability expert team.
-
- Guideline for continued process verification.
-
-
Attachments :
-
-
- Out of trend investigation form issuance logbook. – Attachment-1,
-
- OOT criteria for stability sample. (Formulation) – Attachment-2,
-
- OOT criteria for stability sample (For API /Raw Material) – Attachment-3,
-
- Out of trend investigation form. – Attachment-4,
-
- Guidance for the handling of Human Error. – Attachment-5,
-
- OOT investigation flow for Dissolution. – Attachment-6,
-
- OOT investigation flow for Content uniformity. – Attachment-7,
-
- Out of Trend (OOT) Investigation flow for finished product/raw material/Any other stage sample/stability sample. – Attachment-8,
-
- Out of Trend (OOT) Investigation flow for Blend Uniformity. – Attachment-9,
4.0 Responsibility :
-
-
The analyst shall be responsible for :
-
-
- To immediately inform the Out of Trend (OOT) results to Head QC/section head or designee.
-
- Don’t discard sample solution/stock solution/ instrument settings until the evaluation of Out of Trend (OOT) results.
-
- To participate in the investigation, participate in finding the root cause, and carry out the experimental analysis where applicable.
-
-
Section head or Designee shall be responsible for:
-
-
- To carry out the investigation for Out of Trend (OOT) result as per approved effective SOP’s.
-
- Review historical data of laboratory investigation during the initial assessment to determine if Out of Trend (OOT) have occurred previously, what were the corrective action and preventive action, and the effectiveness of CA & PA.
-
- Interview with the analyst to find out the root cause for Out of Trend (OOT) results.
-
- To review Analytical Test Procedure, Analytical method validation report, Analytical method transfer report, forced degradation data, and discuss with subject matter expert to find the root cause for Out of Trend (OOT) results.
-
- Based on the above review, prepare a hypothesis/simulation study protocol.
-
- Plan hypothesis/simulation study as per approved hypothesis/simulation protocol to find out the root cause or assignable cause for Out of Trend (OOT) results.
-
- To initiate the actions recommended in the Out of Trend (OOT) investigation form.
-
- To derive CA & PA based on the assignable cause identified, perform impact assessment & risk assessment and training.
-
-
Head QC or Designee shall be responsible for :
-
-
- Provide guidance for investigation.
-
- To provide guidance for the hypothesis/ simulation study to find out the root cause for Out of Trend (OOT) results.
-
- Review and approve the hypothesis/ simulation study protocol and report.
-
- Derive CA & PA based on the assignable cause identified, perform impact assessment & risk assessment, and training.
-
- To initiate/monitor the actions recommended as CA and PA in the investigation form.
-
- Review, conclude, and approve the Out of Trend (OOT) reports.
-
- Periodic review of OOT trend and evaluation of the effectiveness of CA and PA derived based on Out of Trend (OOT) Investigation Trend.
-
-
Head QA or designee shall be responsible for:
-
-
- Perform the investigation as per approved effective SOP’s.
-
- Authorize re-sampling as and when required.
-
- Carry out a cross-functional investigation as and when required.
-
- Review and approve the Out of Trend (OOT) investigation form.
-
- Based on the review, if required QA can revise the scope of CA, PA, Impact assessment, and Risk assessment.
-
- Ensure the execution of CA and PA recommended in the Out of Trend (OOT) investigation form.
-
- Periodic trending and review of Out of Trend (OOT) investigation and evaluation of the effectiveness of CA & PA derived based on the OOT investigation trend.
-
-
Head Quality shall be responsible for :
-
-
- To authorize re-sampling as and when required.
-
- Conclude and approve the OOT investigation form.
-
- To ensure the execution of CA & PA recommended in the OOT investigation form.
-
- Intimation to the regulatory department, Analytical Development Department, Formulation Development Department, Corporate Quality, and Customer wherever necessary.
5.0 Definition of terms & abbreviations :
-
-
Assignable cause:
-
-
- A cause that can be attributed as the root cause for the OOT results.
-
- The assignable cause is a conclusion derived from direct or indirect evidence found during the investigation process.
-
-
Cross-Functional investigation:
-
-
- The cross-functional investigation is a process to investigate the OOT thorough review of the manufacturing process, conditions, input materials and it’s documentation as defined in “Event reporting and investigation SOP”
-
-
Hypothesis / Simulation Study:
-
-
- Structured documented Sequence of experiments which is designed to identify the root cause for the failure.
-
- This shall be based on discussions, thought process and scientific rationales’ that what might have gone wrong and resulting in the Out of Trend (OOT) results.
-
- Each Experiment shall have pre-defined expectations that actually what outcome we want to observe through that particular experiment.
-
- Investigational tools can be used to design Hypothesis/Simulation protocol i.e. five whys.?, Cause and Effect Diagram, FMEA, Fish Bone diagram, etc.
-
-
Investigation of Out of Trend (OOT) Result:
-
-
- Review and experiments conducted by an investigator (team of SME’s) and the analyst to find the root cause attributing to Out of Trend (OOT) results.
-
- This could be a Laboratory and/or Cross-functional investigation.
-
- This should be thorough, timely, unbiased well documented, and scientifically proven.
-
-
Out of Trend (OOT):
-
-
- In the case of release analysis, the out of trend results are those results which are within the specification but indicate that the particular analysis or process could be out of control.
-
- In the case of stability out of trend results describes as “A results that are within the specification but do not follow the trend, with respect to previous station results during stability study”.
-
- Note: OOT investigation can be performed for any atypical analytical results or unusual result which is within the specification and Out of Trend (OOT) limits, but need investigation based on the justifiable scientific recommendation from the QA/QC head.
-
-
Out of Specification (OOS):
-
-
- Test results that fall outside the established approved specification limits or acceptance criteria.
-
-
CA (Corrective Action):
-
-
- Action to be taken to correct the cause and avoid the re-occurrence (Reference: ISO guideline).
-
-
PA (Preventive Action):
-
-
- Action to be taken to avoid the occurrence (Reference: ISO guideline).
-
-
Re-test:
-
-
- Testing of Portion of the original sample.
-
- The sample used for the retesting should be taken from the same homogeneous material that was originally collected from the lot, tested, and yielded the Out of Trend (OOT) results.
-
- For a liquid or single-use formulation pack, it may be from the original unit product or composite.
-
- For a solid, it’s to be an additional weighing from the same sample composite prepared for the original test.
-
- This may include the preparation of fresh standards and/ or other test reagents as appropriate (as per approved effective ATP).
-
-
Re-sampling:
-
-
- A new sample from the original container where possible, required in the event of insufficient material remaining from the original sample composite or proven issue with original sample integrity.
6.0 Abbreviations:
-
- CA: Corrective Action.
-
- PA: Preventive Action.
-
- HPLC: High-Performance Liquid Chromatograph.
-
- OOT: Out of Trend.
-
- FMEA: Failure Mode Effect Analysis.
-
- OOS: Out of Specification.
-
- LOD: Loss on Drying.
-
- FDD: Formulation Development Department.
-
- RA: Regulatory Affairs.
-
- ATP: Analytical Test Procedure.
7.0 Procedure for Handling of Out of Trend (OOT) Results:
-
- The purpose of the investigation is to identify the root cause for the Out of Trend (OOT) results. The investigation shall be thorough, timely, unbiased, well documented, and scientifically sound.
-
- If Out of Trend (OOT) results observed, the analyst shall not discard sample solution/stock solution/ instrument settings until the evaluation of OOT results.
-
-
Phase-I: Out of Trend (OOT) Investigation:
-
-
- When OOT results observed, the analyst shall report immediately to the section head or designee.
-
- Section head or designee shall inform to QA immediately and QA person shall take following actions as under,
-
- Issue “Out of Trend Investigation Form” (Attachment-4) to the analyst and make relevant entries in the “Out of Trend Investigation Form Issuance Logbook” (Attachment-1) and “Out of Trend Investigation Form” (Attachment-4)
-
- QA shall issue the OOT form with proper numbering (as per the location numbering system) system.
-
- Section head or designee shall carry out the investigation as per the checklist (but not limited to given in the “OOT Investigation Form”), benchtop audit/on the spot audit, discussion with the analyst, final/stock solutions verification (e.g. Solutions in the same vial/new vial, extra sonication, filter compatibility), as a part of the investigation to find out the root cause for OOT results.
-
- Note: If required based on product history & trend of earlier results, a protocol-based investigational hypothesis study shall be performed to find the root cause for OOT results.
-
-
The section head or designee shall record the investigation finding’s outcome of analyst discussion in the “Out of Trend Investigation Form”.
-
-
- The section head or designee shall mention there comments into the “Summary of investigation” section of the OOT form, based on previous experience of product behavior and trend, raw data checking, discussion with the analyst, final/sock solutions verification, and finding’s shall be reviewed and signed by QA person.
-
- Based on findings (Checklist, raw data review, analyst discussion, benchtop/on the stop audit, final/stock solutions verification),
-
- If assignable cause found then perform the Phase-IA investigation and if the assignable cause is not found then follow the procedure defined in Phase-IB.
-
- Note: For API manufacturing location, the investigation procedure defines for dissolution & content uniformity is not applicable.
-
-
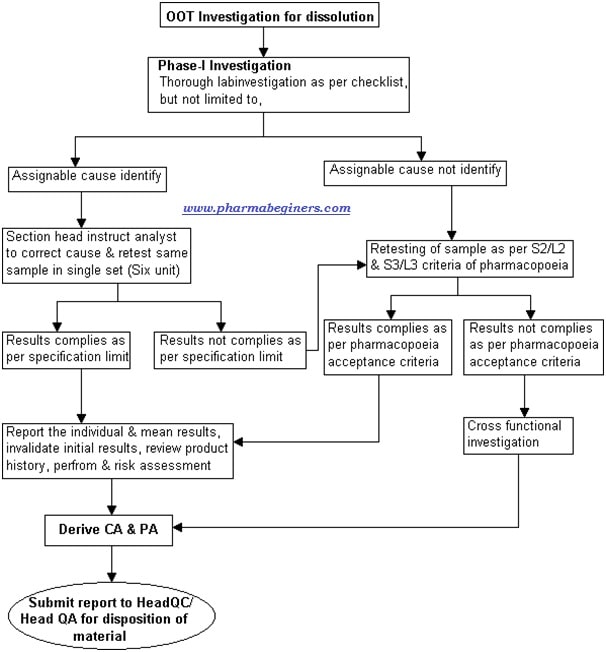
Phase-IA investigation for dissolution:
- Based on the assignable cause identified, the section head or designee shall instruct the analyst to correct the cause and retest the sample in a single set (Six units).
-
-
- Note: Retesting is not required in case of OOT result generated due to reporting error i.e. calculation error/ wrong peak integration /wrong potency used, in such cases correct the cause, report the corrected results, mention the root cause, support by evidence, define the CA & PA and other steps given in OOT form.
-
- Report the retest results into the OOT investigation form and conclude as under,
-
- If results meet the specification limit, invalidate the initial results, review the product history, perform the impact & risk assessment, mention the CA and PA in OOT form and submit the report to Head QC & Head QA/Head Quality for the closing of OOT investigation.
-
- If any individual or mean results do not meet the specification as per pharmacopeia acceptance criteria then proceed for Phase-IB analysis.
-
- Note: Parallel to phase-IB laboratory analysis, intimate to QA designee to initiate a cross-functional investigation.
-
-
Phase-IB analysis for dissolution:
-
-
- If no laboratory error identified or results do not comply in Phase-IA investigation, perform further analysis as under,
-
- Analyze the sample as per S2/S3 & L2/L3 (As per USP general chapter <711>) stage criteria of pharmacopeia and report the results.
-
- Evaluate the results based on criteria provided in the pharmacopeia.
-
- The final decision of batch release shall be made by Head QA/Head Quality, based on the outcome of cross-functional investigation & reported results of laboratory analysis.
-
- If at any stage of investigation results confirm that the dissolution result qualifies for the OOS, initiate the OOS investigation as per OOS guideline (from Phase-IB).
-
- In case of hard or soft gelatin capsules or gelatin-coated tablets, dissolution results do not comply against approved effective specification then retest sample as per S1/S2/S3 criteria of pharmacopeia as under,
-
- Where water or a medium with a pH of less than 6.8 is specified as the medium in the individual analytical test procedure, the same medium specified shall be used with the addition of purified pepsin that results in an activity of 750,000 units or less per 1000 ml and for media with a pH of 6.8 or greater, pancreatin can be added to produce not more than 1750 USP units of protease activity per 1000 ml (Refer USP general chapter <711>).
-
-
Report the retest results in out of trend investigation form.
-
-
- Note: In COA it shall be mentioned that results comply in pancreatic media.
-
- Results shall be evaluated based on criteria provided in the pharmacopeia.
-
- The final decision of batch release shall be made by Head QA/Head Quality, based on the outcome of cross-functional investigation & reported results of laboratory analysis.
-
- Note: If in any dissolution Out of Trend (OOT) (for stability samples), the cross-functional investigation is performed for the earlier station then Head QA/Head Quality can take the decision for not performing further cross-functional investigation,
-
- This decision shall be supported by the scientific rationale and shall be documented in the current OOT form.
-
-
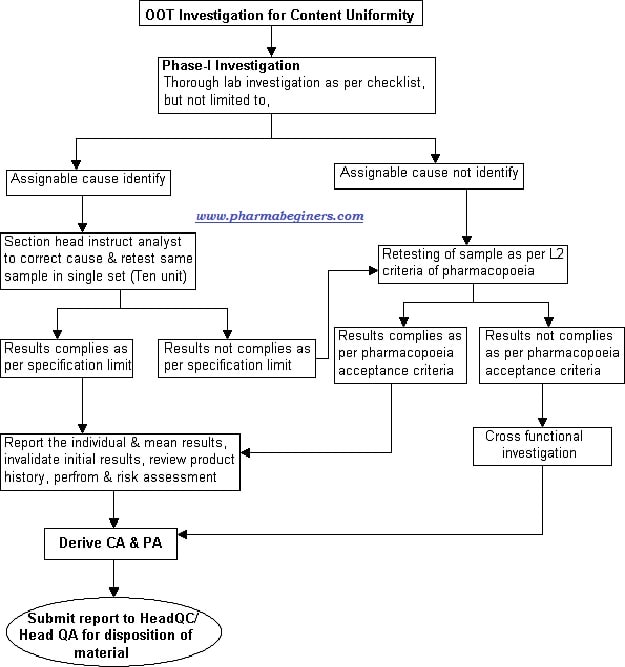
Phase-IA investigation for Content uniformity:
-
-
- Based on the assignable cause identified, the section head or designee shall instruct the analyst to correct the cause and retest the sample in a single set (10 units).
-
- Note: Retesting is not required in case of Out of Trend (OOT) result generated due to reporting error i.e. calculation error/ wrong peak integration /wrong potency used, in such cases correct the cause, report the corrected results and mention the root cause, CA & PA and other steps given in OOT form.
-
- Report the retest results into the Out of Trend (OOT) investigation form and conclude as under,
-
- If results meet the specification limit, invalidate the initial results, review the product history, perform the impact & risk assessment, mention the CA and PA in Out of Trend (OOT) form and submit the report to Head QC & Head QA/Head Quality for the closing of OOT investigation.
-
- If any individual or mean results does not meet the specification as per pharmacopeia acceptance criteria then proceed for Phase-IB investigation (step no. 6.3.6).
-
- Note: Parallel to phase-IB laboratory analysis, intimate to QA designee to initiate a cross-functional investigation.
-
-
Phase-IB investigation Content Uniformity:
- If no laboratory error identified or results do not comply in Phase-IA investigation, perform further analysis as under,
-
-
- Analyze the sample as per L2 (As per USP general chapter <905>) stage criteria of pharmacopeia and report the results.
-
- Evaluate the results based on criteria provided in the pharmacopeia.
-
- The final decision of batch release shall be made by Head QA/Head Quality, based on the outcome of cross-functional investigation & reported results of laboratory analysis.
-
-
Identification of Out of Trend (OOT) results in Finished Product/Raw material/ Any other sample & Stability sample) :
-
-
- Out of Trend (OOT) results in finished products, raw materials, any other stage samples & stability samples shall be identified as under,
-
- Identification of Out of Trend (OOT) in Finished Product/ Raw material/ any other stage sample: Out of Trend (OOT) results in the finished product, raw material, any other sample shall be identified through current version SOP on “Continued process verification and OOT results shall be confirmed by following laboratory investigation procedure.
-
- Identification of Out of Trend (OOT) instability Sample: OOT criteria for formulation manufacturing location is defined in attachment-2 & for API manufacturing location define in attachment-3.
-
- After identification of Out of Trend (OOT), further laboratory investigation shall be performed to confirm that whether OOT results associated with laboratory error or manufacturing process related.
-
- Out of Trend (OOT) results in investigation procedure for finished product/Raw material/ other stage sample & stability sample:
-
- When Out of Trend (OOT) results are identified, then further laboratory investigation shall be performed to confirm the OOT results as under,
-
- Section head or designee shall perform the preliminary investigation as per procedure defined & conclude as under,
| S.No. | Observation |
Investigation stage |
|
1 |
If assignable cause observed based on a checklist, Raw data review, analyst discussion, final/stock solutions verification (e.g. Solutions in the same vial/new vial, extra sonication, filter compatibility) | If assignable cause found, follow the investigation procedure define in Phase-IA. |
|
2 |
If no assignable cause found checklist, raw data review, analyst discussion, final/stock solutions verification (e.g. Solutions in same vial/new vial, extra sonication, filter compatibility). | Follow the investigation procedure define in Phase-IB. |
-
-
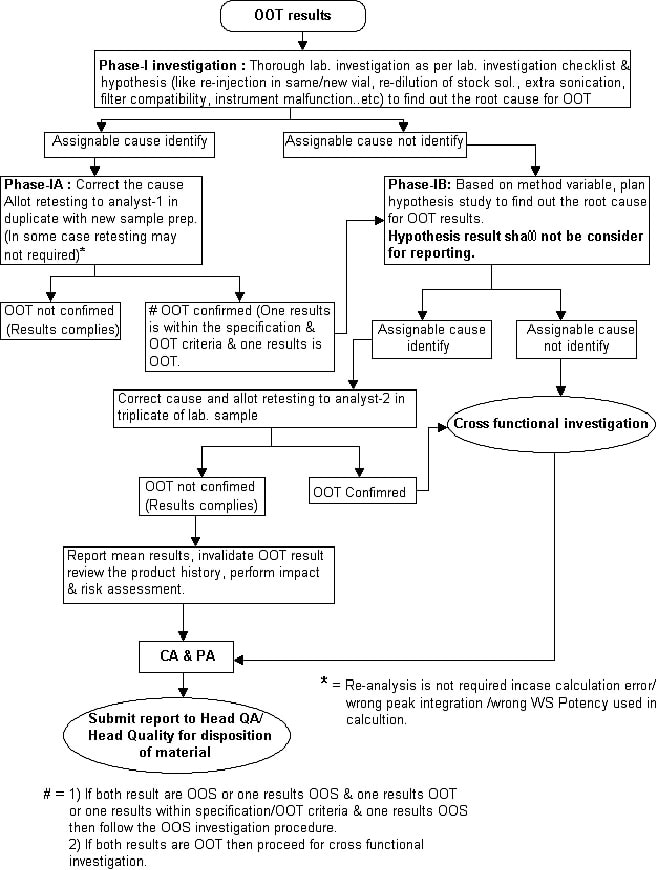
Phase-IA investigation (If the assignable cause is found based on a checklist, raw data review/ final/stock solutions verification):
-
-
- Based on the assignable cause identified, the section head or designee shall instruct the analyst to correct the cause and retest the sample in duplicate (two new sample preparation) with the same sample pool.
-
- Note: Retesting is not required in case of OOT results generated due to reporting error i.e. calculation error/ wrong peak integration / wrong potency used, in such cases correct the cause and report the corrected results and mention the root cause, CA & PA, and other steps given in OOT form.
-
- Report the retest results into the OOT investigation form and conclude as under,
-
- If results meet the specification limit and within the OOT limit then invalidate the initial results, review the product history, perform the impact & risk assessment, mention the CA and PA in OOT form and submit the report to Head QC & Head QA/Head Quality for the closing of OOT investigation.
-
-
If the result is OOS or OOT, performed further investigation as under,
-
| S.No. | Observation |
Investigation stage |
| 1 | If both results are out of specification. | Perform investigation as per OOS investigation (Initiate from Phase-IB) |
| 2 | If one result is OOS &second results are within specification &OOT limit. | Perform investigation as per OOS investigation (Initiate from Phase-IB) |
| 3 | If one result is OOS &second result is within specification limit but OOT. | Perform investigation as per OOS investigation (Initiate from Phase-IB) |
| 4 | If one result is within specification &OOT limit & second result is OOT. | Follow phase-I B investigation as per the OOT guideline (step no. 6.5.4). |
| 5 | If both results are OOT and within specification. | Intimate to QA for cross-functional investigation. |
-
- Phase-IB (If the assignable cause is not found based on checklist/initial hypothesis study) extended lab. investigation:
-
- Hypothesis/Simulation study protocol shall be designed in discussions with, analysts, SME’s and reviewers.
-
- The analytical procedure defined in ATP’s and analysis performed by the analyst (based on the interview of analyst) shall be discussed in detail to find out the variables which can lead to OOT results.
-
- Section head or designee shall plan the hypothesis/ simulation (like sonication temperature increase or decrease, wrong solvent/ reagent used, glassware contamination, based on variables identified through above study through the approved protocol to find out the root cause for OOT
-
- Experiments planned in hypothesis/simulation protocol shall have predefined.
-
- Do not use the investigation testing results (hypothesis/ simulation study results) to disregard original OOT results. It is only to confirm or eliminate a probable root cause.
-
- Section head or designee shall review the hypothesis/ simulation study report and if assignable cause identify based on the hypothesis/simulation study, shall recommend for retesting of the sample as under,
-
-
Section head or designee shall allot the sample for retesting in duplicate to the analyst with new sample preparation.
-
-
- Report the retest results into the OOT investigation form and conclude as under,
-
- If all results (individual & mean results) comply with the specification limit, invalidate the initial results, review the product history, perform impact & risk assessment, derive CA & PA and submit the report to Head QC and Head QA/Head Quality for the closing of OOT investigation.
-
- Any result (individual or mean results) confirms the OOT criteria, then intimate to Head QA or designee to initiate a cross-functional investigation.
-
- The final decision of batch disposition shall be decided by Head QA/Head Quality based on the outcome of the cross-functional investigation, review of phase-IB results, review of product history, other test & stages results.
-
-
General Procedure for Handling of Out of Trend (OOT) Results:
-
-
- After completion of OOT investigation/cross-functional investigation, Head QA or designee shall ensure the necessary attachments filed with the OOT investigation form.
-
- After review of the OOT investigation/ cross-functional investigation, CA & PA (As per respective plant SOP) form shall be issued by QA and ensure compliance of CA & PA.
-
- The impacted department Head shall monitor each recommendation mentioned in the OOT form.
-
- If OOT investigation requires more time for closing then the defined period.
-
- Prepare the interim report for the extension of closure. Extension of closure shall have proper justification and reason for the extension.
-
-
It shall have the brief “As on date status” for the investigation.
-
-
- Allowed the extension for one month from the date of the request for the extension.
-
- Document every step and preserve the records as a part of the lab.
-
- Impact assessment and risk assessment an integral part of the OOT investigation form.
-
- Even if a batch is rejected based on an OOT result, the investigation is necessary to establish the root cause. Impact assessment of the established root cause shall be performed with respect to other manufacturing batches. Batch rejection does not negate the need to perform the investigation for the root cause establishment.
-
- In case the solution is not stable then perform further investigation by new sample preparation with appropriate justification.
-
- At any stage, perform further investigation (re-opened) based on scientific justification/rationale.
-
- At any stage, if the sample is not available for further investigation, Carryout the re-sample with proper justification to performed further investigation.
-
- Human errors are identified as root cause then handle the human error as per attachment-5 to derive CA & PA.
-
-
Trending and Evaluation of OOT:
-
-
- Trending and evaluation of OOT shall be carried out at quarterly basis by QA.
-
- QA shall review the data and evaluate the repetitive type OOT as per following but not limited to,
-
-
- 1) Repetitive OOT in a particular test of the product
-
-
-
- 2) Repetitive OOT by particular analyst
-
-
-
- 3) Repetitive OOT of any stage of product sample etc.
-
-
-
- 4) Repetitive OOT on the particulate instrument.
-
-
-
- 5) Repetitive OOT due to Human Error.
-
-
- QA shall conclude the repetitive OOT from the OOT trend and shall derive CA & PA.
-
- Head QC or designee shall assess the effectiveness of CA & PA, derived based on OOT trends.
Attachments :
Attachment 1 – Out of trend investigation form issuance logbook.
Make the logbook table (tabulated form) with the following contents.
-
- Sr. No.
-
- Product /Material Name
-
- Batch No. / AR No.
-
- Stage
-
- Test Parameter
-
- OOT Form No.
-
- Details of OOT Investigation Form
-
-
- Issued to
-
-
-
- Issued by (Sign/Date)
-
-
-
- Received by (Sign/Date)
-
-
- Status of OOT Investigation (Open / Closed)
-
- Incident No. (If any)
-
- Root Cause
-
- CAPA No. (If any)
-
- Remark (If any)
Attachment-2 – OOT criteria for stability sample. (Formulation) :
Assay: If the difference in assay results observed more ± 3.0% from the previous stability station &± 5.0% from the initial stability station.
Dissolution: If the difference of mean value observed ± 10% from the previous stability station. Note: For the dissolution profile these criteria applicable for every time point.
Water/LOD test (Applicable only if results are about 1.0%): Absolute difference of water content/LOD observed ± 0.5 from the previous stability station.
Related substance
-
- When the specification limit is up to 0.20%: If increase or increase in results ± 50% from previous stability station and the difference in results between previous ¤t stability station is >0.05%.
-
- When the specification limit is more than 0.20%.: If a decrease or increase in results ± 50% from previous stability station and the difference in results between previous ¤t stability station is >0.1%.
Preservative content: If the difference in results observed more ± 5.0% from the previous stability station.
Attachment-3- OOT criteria for stability sample (For API /Raw Material) :
Water/LOD test (Applicable only if the result is about 1.0%): Absolute difference of water content/LOD observed ± 0.5 from the previous stability station.
Related substance :
-
- When the specification limit is up to 0.20%.: If the increase or increase in results ± 50% from previous stability station and the difference in results between previous ¤t stability station is >0.05%.
-
- When the specification limit is more than 0.20%.: If a decrease or increase in results ± 50% from previous stability station and the difference in results between previous ¤t stability station is >0.1%.
Attachment-4- Out of trend investigation form.
Click for Download – OOT-Attachment 4
Attachment-5 – Guidance for the handling of Human Error.
A) Error – Knowledge Based
-
- Corrective Action
-
-
- Procedural requirements
-
-
-
- Train Employee
-
-
- Preventive Action
-
-
- Review Department Training Program
-
-
-
- Close Gaps
-
B) Error – Lapse Based
-
- Corrective Action
-
-
- Counsel Employee (Verbal, Written, Disciplinary)
-
-
- Preventive Action
-
-
- GMP training
-
C) Error – Cognitive Based
-
- Corrective Action
-
-
- Match Employee to Abilities
-
-
- Preventive Action
-
-
- Evaluate Knowledge Assessment Effectiveness
-
D) Error – Value-Based
-
- Corrective Action
-
-
- Counsel Employee (Verbal, Written, Disciplinary)
-
-
- Preventive Action
-
-
- GMP training
-
E) Error – Skill Based
-
- Corrective Action
-
-
- Match Employee to Abilities
-
-
- Preventive Action
-
-
- Evaluate Knowledge Assessment Effectiveness
-
F) Error – Time Pressure
-
- Corrective Action
-
-
- Assessment of resources
- Reassign duties
- Create or Update Plans (Planning)
-
-
- Preventive Action
-
-
- Conduct process FMEA
-
G) Error – Inadequate Equipment
-
- Corrective Action
-
-
- Increase Inspection & Checks
- Equipment Lock Out / Tag Out
- Re qualify / Revalidate after fixing or replacing Equipment
-
-
- Preventive Action
-
-
- Evaluate the adequacy of Qualification / Validation Resources and Procedures
-
-
-
- Close Gaps
-
H) Error – Fatigue
-
- Corrective Action
-
-
- Assessment of resources
- Plant layout study
- Reassign Duties
-
-
- Preventive Action
-
-
- Flow chart Processing
-
-
-
- Conduct Process FMEA
-
-
-
- Evaluate Resources Lading
-
-
-
- Close Gaps
-
I) Error – Inexperience
-
- Corrective Action
-
-
- Match Employee to Abilities
-
-
- Preventive Action
-
-
- Evaluate Knowledge Assessment Effectiveness
-
J) Error – Design & Construction Deficiencies
-
- Corrective Action
-
-
- Increase Inspection &Checks
- Equipment Lock Out / Tag Out
- Re qualify / Revalidate after fixing or replacing Equipment
-
-
- Preventive Action
-
-
- Evaluate the adequacy of Qualification / Validation Resources and Procedures.
-
-
-
- Close Gaps
-
K) Error – Unworkable Procedures
-
- Corrective Action
-
-
- Revise Procedure
- Conduct Training to the new revision
-
-
- Preventive Action
-
-
- Flow chart Processing
-
-
-
- Conduct Process FMEA
-
-
-
- Update procedure
-
-
-
- Conduct Training
-
Attachment-6 – OOT investigation flow for Dissolution.
Attachment-7 – OOT investigation flow for Content uniformity.
Attachment-8 – Out of Trend (OOT) Investigation flow for finished product/raw material/Any other stage sample/stability sample.
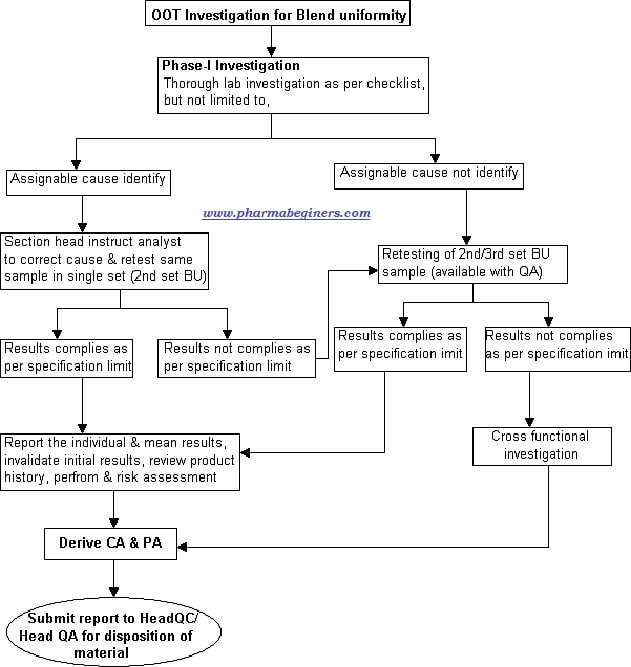
Attachment-9 – Out of Trend (OOT) Investigation flow for Blend Uniformity.



Pingback: SOP - Corrective Action and Preventive Action (CAPA) - Pharma Beginners