Aseptic technique is a critical requirement for collecting and testing sterile and non-sterile samples in order to avoid contamination that could provide incorrect test results.
SOP for Aseptic Technique
1.0 OBJECTIVE :
-
- The objective of this Standard Operating Procedure (SOP) is to define proper aseptic a technique during microbiological sampling and testing to avoid contamination and assure accurate test results.
2.0 SCOPE :
-
- Aseptic technique is a critical requirement for collecting and testing sterile and non-sterile samples in order to avoid contamination that could provide incorrect test results.
-
- This standard provides guidance for the proper aseptic technique for
-
-
- The sampling of Product,
-
-
-
- In-process samples,
-
-
-
- Raw Materials,
-
-
-
- Primary Packaging Material,
-
-
-
- Utility systems,
-
-
-
- Collecting validation samples,
-
-
-
- Performing environmental monitoring and
-
-
-
- Personnel Monitoring.
-
-
- Guidance is also provided for proper aseptic technique when preparing for and performing tests and analyses in the laboratory, including standard laboratory tests, such as
-
-
- Dilutions,
-
-
-
- Plate Counts,
-
-
-
- Water Testing,
-
-
-
- Microbial Enumeration Test
-
-
-
- Test for Specified Pathogens,
-
-
-
- Sterility Testing,
-
-
-
- Endotoxin Determination,
-
-
-
- Culturing of Biological Indicators,
-
-
-
- Bioassays and the other testing.
-
-
- The aseptic technique described herein is provided as a foundation to be practiced with all microbiological sampling and testing to prevent contamination of test samples and to minimize the possibility of mixed (contaminated) cultures.
3.0 RESPONSIBILITY – ASEPTIC TECHNIQUE :
-
-
Head Microbiology / Designee shall be responsible for:
-
-
- Assuring that all personnel has been trained and qualified in proper aseptic technique before being assigned activities that require collecting or testing any material.
4.0 ABBREVIATIONS AND DEFINITIONS -ASEPTIC TECHNIQUE :
-
-
Aseptic :
-
-
- Absence of viable microorganisms
-
-
Aseptic Filling :
-
-
- Part of aseptic processing where a pre-sterilized product is filled and/or packaged into sterile or dehydrogenated containers and partially closed and/or closed.
-
-
Aseptic Processing :
-
-
- Assembly of sterilized components and products in a controlled environment, in which the air supply, materials, equipment, and personnel are regulated to control microbial and particulate contamination to acceptable levels.
-
-
Aseptic Processing Area :
-
-
- An area that has defined environmental control of particulate and microbial contamination and is constructed and used in such a way as to reduce the introduction, generation, and retention of contaminants within the area used for the processing of sterile products.
-
-
Aseptic Process Simulation (APS) :
-
-
- A means for establishing the capability of an aseptic process, as performed using a growth medium in place of the typically filled material.
-
- Note that APS is understood to be synonymous with media fill.
-
-
Growth Promotion Test :
-
-
- Test performed to demonstrate that media will support microbial growth per Pharmacopeia specified challenge organisms.
-
-
Intervention :
-
-
- An aseptic manipulation or activity performed by personnel that occurs within the critical area.
-
-
Sanitization :
-
-
- The act or process, physical or chemical, of reducing viable organisms on a surface to a defined acceptable level.
-
- Sterile: Absence of any viable organisms.
-
- Sterility Test: Tests performed to determine if viable microorganisms are present.
-
- Sterilization: The act or process, physical or chemical, of destruction or elimination of all viable organisms.
5.0 Process Flow – Aseptic Technique :
-
- Work Flow -1
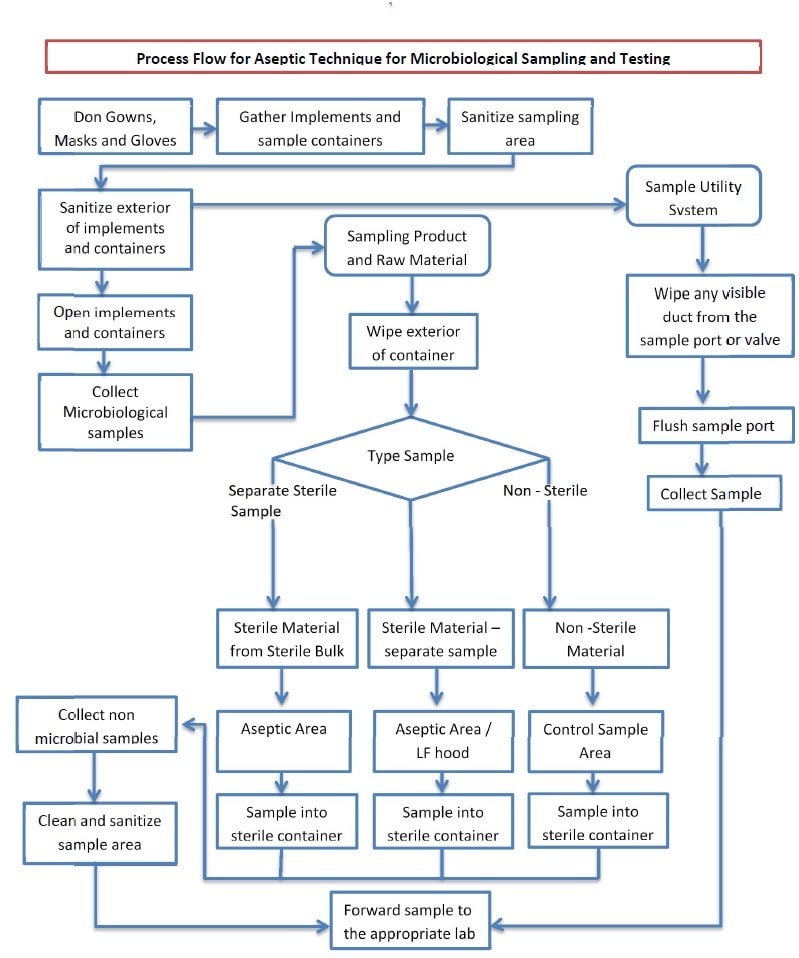
-
-
Work Flow -2
-
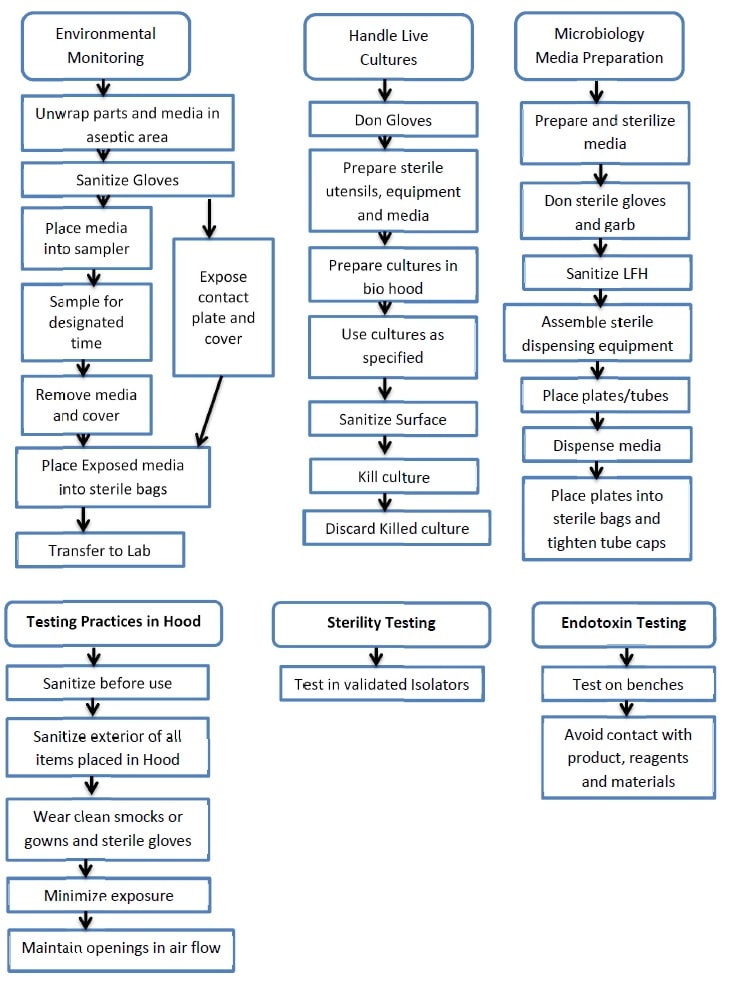
6.0 PROCESS DESCRIPTION – ASEPTIC TECHNIQUE :
-
-
General Sampling Process under the Aseptic Technique :
-
-
- Sample documentation procedures shall be followed for intimations and sample processing (refer to the current version of SOP for Microbiological Culture Management.
-
- Sampling for microbiological testing shall be performed prior to collecting samples for other purposes.
-
- Sampling personnel shall wear clean, lint-free smocks, gowns or uniforms specified for the area where sampling is to occur.
-
- Sterile gloves shall be worn and sanitized just before sampling.
-
- Non-sterile material shall be sampled under controlled environments, such as a Laminar Air Flow hood or room with HEPA filtered air.
-
- All microbiological samples shall be collected using clean, sterile, pyrogen-free implements, such as forceps, scoops, pipettes, hoses and sample thieves.
-
- Utility system samples shall be collected from existing hoses attached to the ports, if available.
-
-
Sanitize sampling tables and hoods using a validated sanitizing agent before use.
-
-
- Sanitize the exterior of the sampling implement packages and containers with a validated sanitizing agent prior to use with a clean lint-free wipe/cloth.
-
- Sanitization of the item/system to be sampled shall depend upon sample type, such as utility system, raw materials, bulk product, environmental monitoring plates and swabs, etc.
-
- Open packages and sample containers just before collecting the sample in order to minimize exposure to the environment.
-
- Sample container lids shall be held such that the interior surface of the lid faces downward to avoid contamination falling into it.
-
- If the sample container lid cannot be held while sampling, it may be placed on a sanitized/sterilized surface with the interior lid surface facing down.
-
- Microbiological samples for aseptic manufacturing facilities shall be collected into sterile sample containers when used for testing other than the endotoxin test or endotoxin-free sample containers when the endotoxin test is to be performed.
-
- Microbiological samples for non-sterile dose facilities shall be collected into sterile sample containers.
-
- Gloves shall be changed between samples or sanitized with an approved sanitizer and allowed to dry.
-
- Sampling areas shall be cleaned and sanitized after each sample is taken and after completing sample collection.
-
- Transport samples in a covered container to the appropriate Microbiology/Environmental Monitoring Laboratory, as soon as sampling is completed.
-
-
Sampling Product and Raw Material (Aseptic Technique):
-
-
- Wipe off the exterior of material containers with a validated sanitizing agent before opening.
-
- Ensure that the required contact time for the approved sanitizer has been achieved.
-
- Non-sterile raw materials and components, such as stoppers, caps, bottles, etc., shall be sampled in an enclosed environment that is temperature- and humidity-controlled.
-
- Sample tables, carts, scales, and booths shall be cleaned and sanitized prior to collection of samples.
-
- Avoid leaning over open containers.
-
- Sterile raw materials and components, such as sterile plastic containers, droppers, caps, stoppers, etc., shall be sampled in a sanitized laminar airflow hood in an environmentally controlled, cleanroom.
-
-
- Typically, separate representative samples are provided by the vendor for testing purposes, so it is not generally necessary to sample from bulk material containers.
-
-
-
- If sampling from sterile bulk materials is required it shall only be performed in an aseptic sampling area.
-
-
-
- Sterile samples are to be placed into sterile sample bags or bottles.
-
-
-
Sampling Utility Systems (Aseptic Technique):
-
-
- Utility system sampling requires additional care because samples are collected in controlled and uncontrolled areas of the facility.
-
- Sampling personnel shall wear clean smocks or gowns and don sterile gloves before collecting any sample.
-
- Sampling apparatus and containers shall be clean, sterile, and/or endotoxin-free as per test requirements.
-
- Wipe any visible dust from the exterior of the sample port or valve using a lint-free wipe/cloth moistened with an approved sanitizer.
-
- Do not spray or wipe the interior of the port or valve.
-
- Ensure that the required contact time for the sanitizer has been achieved.
-
- If a hose is already connected to the port or valve, collect the sample directly from that hose.
-
- If the hose is not connected but is provided, attach the hose using procedures applicable for that system. Replace the used hose onto the provided clamp after sampling so that it fully drains.
-
- If a hose is not present, a sterilized adapter and hose may be used to sample directly from the valve/port.
-
- Flush the sample port/valve for the qualified time or volume and immediately open the labeled sample container and collect the sample.
-
- Close the container immediately and record the sampling time.
-
-
Environmental Monitoring (Aseptic Technique):
-
-
- Unwrap sterilized environmental monitoring parts, equipment, and media only in the aseptic processing area.
-
- Sanitize gloves before handling any sterilized parts, equipment, and media.
-
- Swab tubes and the bottom of agar media plates shall be labeled with sample site information before exposing them.
-
- Open the contact plate, roll media onto the surface to be sampled and immediately replace the lid.
-
- Place exposed plates into sterile bags or sanitized trays and transfer to the laboratory for further processing.
-
- If swabbing is to be performed in place of contact plates, carefully remove the moistened swab from the specified tube, swab the area or item as defined in the procedure or protocol and immediately place back
in the swab tube.
- If swabbing is to be performed in place of contact plates, carefully remove the moistened swab from the specified tube, swab the area or item as defined in the procedure or protocol and immediately place back
-
-
Transfer swabs to the laboratory for further processing.
-
-
- Handle air sampler parts by the outside, non-sterile sides/edges, being careful not to touch the air inlet port.
-
- When placing plated media into air samplers, open the plate and immediately place into the sampler, close the cover and start sampler.
-
- Place media plate lid, interior surface downward on a sanitized surface close to the sampler.
-
- At the completion of the sampling period, remove media from the sampler, and immediately cover the plate.
-
- For exposure using settle plates, the settling plate shall be opened at the point of sampling by holding the lid from the edges and the lid shall be placed near to the Petri plates.
-
- After completion of the sampling duration, the plate shall be covered using the same lid and care shall be
taken that the plate is not accidentally exposed again.
- After completion of the sampling duration, the plate shall be covered using the same lid and care shall be
-
- Do not contact the surface of any media with gloved hands.
-
- If the media is accidentally contacted, discard it and use another plate.
-
- Place all exposed plates into sterile bags/containers for transfer to the laboratory for incubation.
-
-
Handling of Live Cultures – Aseptic Technique :
-
-
- Live cultures include growth promotion challenge cultures and dried pellets, contaminated environmental monitoring plates, positive controls, positive biological indicators, microbial identification cultures, etc.
-
- All live cultures shall be segregated from areas used for sample testing and optimally, handled in a different area of the laboratory within a Biosafety Cabinet.
-
- Positive control dilutions and inoculation shall be performed in a biological safety hood/cabinet.
-
- Agar plates containing fungal cultures shall be sealed with parafilm to prevent the spread of spores.
-
- Surfaces in areas where a live culture plate, tube, bottle, pellet, etc., was opened shall be sanitized immediately after use by using an approved sanitizer for the appropriate contact time.
-
- Gloves shall be used when working with live cultures.
-
- Live cultures can be contaminated by poor aseptic technique.
-
- To maintain the purity of the culture, sterile utensils, glassware and materials shall be used for transfers, dilutions, plate counting, etc.
-
- Staff engaged in sampling or sample analysis activities shall not enter/work in live culture handling area or vice versa unless clothing and gowns are changed and hands are thoroughly cleaned with sanitized.
-
- Staff engaged in live culture work shall not enter aseptic manufacturing or sterility test areas until they exit the facility and reenter per personnel entry procedures for hygiene and dress.
-
- Live cultures shall be killed per validated decontamination cycle before discarding.
-
-
Media Preparation under the Aseptic Technique :
-
-
- The media prepared in-house should be subjected to a validated stream sterilization cycle to ensure that it is free from viable microorganisms.
-
- Once sterilization has been completed, agar can be cooled to 45-50°C to minimize condensation and provide for easier handling while dispensing.
-
- The media prepared in-house requires proper aseptic technique to dispense sterilized media into plates and tubes.
-
- Operators dispensing media shall wear sterile gloves and a clean gown with sleeve covers.
-
- Media dispensing shall be performed under a sanitized laminar flow hood using sterilized dispensing equipment and utensils.
-
- The media may be poured manually from the sterile media container after the outside of the container has been wiped with sanitizer.
-
- Wipe the exterior of the plate or tube package with validated sanitizer and place it into the hood.
-
- Open packages and place plates or tubes onto the hood work surface.
-
- Sanitize gloves between each operation.
-
- Dispense media into plates/tubes and avoid contact with media.
-
- After solidification, tighten screw caps on tubes and ensure that plates are covered properly.
-
- Place plates into sterile bags or containers/trays for storage.
-
- Prepared plates and tube media made in-house or purchased from a vendor, shall be incubated prior to use to assure there is no contamination prior to use.
-
- Samples of media poured directly into plates during plate counting or other testing may be incubated concurrently with the test as negative controls.
-
-
Testing Practices in Laminar Flow and Biological Safety Cabinet (Aseptic Technique):
- Approved sanitizing agents shall be used to wipe down the walls and benchtop before using laminar flow or biological safety cabinets and allowed to remain for the contact time prescribed for the sanitizing
agent.
-
-
- The exterior surfaces of all materials, parts, samples, and equipment placed into the hood shall be sanitized with an approved agent prior to beginning tests.
-
- Personnel shall wear clean smocks or gowns and sterile gloves when testing within hoods.
-
- Gloves shall be changed if hands are removed from the hood/cabinet and contact non-sanitized items outside of the hood.
-
- Gloves shall be sanitized again when hands are placed within the hood.
-
- Minimize the time samples and media containers are open under the hood/cabinet by opening a container just before collecting a sample or inoculating media and closing the container immediately after use.
-
- Assure that open container are within the clean airflow.
-
- Do not overcrowd the hood/cabinet workbench while working.
-
- Place material near/closer to the horizontal laminar airflow hood filter face or to the rear of vertical biological safety cabinets.
-
-
STERILITY TESTING – ASEPTIC PRACTICES:
-
-
- Sterility testing shall be performed in validated Grade “A” Laminar Flow Hoods within Class C clean rooms or in validated, sterilized isolators to prevent contamination of media and test samples.
-
- The same aseptic gowning and personnel aseptic technique used for manufacturing sterile product shall be followed when performing sterility testing in hoods.
-
- When testing under Laminar Flow Hoods, requirements in above sections shall also be followed, with the exception that aseptic gowning is required for sterility testing.
-
- Live cultures shall not be permitted where sterility testing is performed.
-
-
ENDOTOXIN TESTING – ASEPTIC TECHNIQUE:
-
-
- Endotoxin testing is generally performed on laboratory benches, but good aseptic technique is required to avoid contamination of the sterile reagents and product, as well as endotoxin-free pipettes and tubes that
are used to make dilutions and inoculate lysate with samples.
- Endotoxin testing is generally performed on laboratory benches, but good aseptic technique is required to avoid contamination of the sterile reagents and product, as well as endotoxin-free pipettes and tubes that
-
-
BIOASSAYS – ASEPTIC TECHNIQUE:
-
-
- Proper aseptic technique shall be used during the collection of bioassay samples and during testing to prevent contamination of the sample that could adversely influence Bioassay results.
-
- Sterile sample bottles shall be used to collect Bioassay samples.
-
- Sterile reagents and implements used to perform microbial assays of antibiotics and vitamins shall be sterile.
-
- Bioassays shall be performed under laminar flow hoods using the aseptic technique described in above sections.
-
- Aseptic technique for handling live cultures and media for bioassays are described in above sections.
-
- Aseptic technique unique to the specific bioassay shall be detailed in the validated methods.
-
-
REQUIREMENTS OF ASEPTIC TECHNIQUE:
- All personnel assigned to sample or test material for microbiological quality shall be trained in proper aseptic technique before assignment to any related task.
-
-
- Each Micro lab shall develop written procedures for sampling and testing microbiological samples that include all requirements for personnel and material protection from contamination.
-
- Each Micro lab shall procure appropriate sampling devices, locations, and hoods to allow for the aseptic collection of microbiological samples:
-
-
- Non-sterile raw materials and components shall be sampled in an enclosed environment that is temperature- and humidity-controlled.
-
-
-
- Sterile raw materials and components shall be sampled in a sanitized laminar airflow hood in an environmentally controlled and monitored aseptic cleanroom.
-
-
-
- Implements and sample containers may be purchased as one-time-use sterile packages or may be cleaned and sterilized/dehydrogenated in-house per validated processes.
-
-
- Each Micro lab shall provide clean smocks or gowns, masks, and sterile gloves for personnel collecting and testing microbiological samples.
-
- Sanitizing agents shall be validated for their intended use during sampling and testing.
-
- If sterile bulk material containers must be sampled, the Micro lab shall develop a written procedure Training Program to protect the sterile material from contamination.
-
- This sampling process shall also be included in Aseptic Process Simulations.
-
- Gloves shall be changed or sanitized with an approved sanitizer between samples.
-
- The best practice for endotoxin testing is to change gloves, since a sanitizer may render gloves sanitary, but may not be effective in removing endotoxin contamination.
-
- The procedure shall include the process for entering and working in the sample areas, including personnel hazards, and required personal protective equipment.
-
-
Written procedures shall be developed for sampling each utility system.
- Environmental monitoring personnel shall be trained and qualified to gown and to enter and work in aseptic processing areas per the training standard for aseptic processing areas.
-
-
- Microbiology Lab shall have procedures in place for handling live microbial cultures, including
-
-
- Growth promotion challenge cultures and dried pellets,
-
-
-
- Contaminated environmental monitoring plates and media,
-
-
-
- Positive controls,
-
-
-
- Positive biological indicators,
-
-
-
- Microbial identification cultures,
-
-
-
- Aseptic technique for streaking for purity, etc.
-
-
- Provide adequate space in Microbiology Laboratories, so that live cultures are separated from other laboratory areas in order to prevent contamination of samples and clean sample testing areas.
-
- Provide adequate space and validated sterilization equipment for preparation of microbiological media.
-
- Plant shall provide sufficient space for the required personnel to move about the laboratory without the undue chance of interfering/contaminating the work of others.
-
- Provide separate storage and processing areas for contaminated samples, media, and supplies.
-
- Separate decontamination equipment shall be available for decontamination of materials and
supplies prior to disposal.
- Separate decontamination equipment shall be available for decontamination of materials and
-
- Each Micro lab shall validate the sterilization and decontamination cycles.
-
- Microbiology personnel involved with Aseptic Process Simulations shall be trained in aseptic technique.
-
- Personnel working in laminar flow or biological containment hoods shall be trained in aseptic technique and shall be familiar with airflow in the hood in order to avoid blocking clean HEPA filtered airflow from flowing over open sterile material.
-
- Equipment, supplies and operator extremities shall be maintained downstream of open sterile material.
-
- Each Micro lab shall provide sterility testing facilities maintained under the same environmental conditions as manufacturing aseptic processing facilities.
-
- These facilities may be either validated Grade “A” Laminar Flow Hoods within Grade “C” rooms or validated isolators and associated transfer and sterilization equipment.
9.0 REFERENCES – ASEPTIC TECHNIQUE :
-
- FDA Guidance for Industry: Current Good Manufacturing Practice – Sterile Drug Products Produced by Aseptic Processing.
-
- Parenteral Drug Association – Fundamentals of an Environmental Monitoring Program
-
- Pharmaceutical Inspection Convention and Pharmaceutical Inspection Cooperation Scheme (PIC/S): Guide to Good Manufacturing Practice for Medicinal Products
-
- USP General Chapter <1116> Microbiological Evaluation of Clean Rooms and Other Controlled Environments
-
- USP General Chapter <1117> Microbiological Best Laboratory Practices
*********************************************END*********************************************



Pingback: Media Fill Validation Procedure & Guideline - Guidelines - SOPs
Pingback: Microbiological Analysis of Material & Drug Product - Pharma Beginners
Pingback: Microbiological Media Management - SOP & Guideline - Pharma Beginners
Pingback: Growth Promotion Test and Inhibition Test of Media - Pharma Beginners